ISHLT 46th Annual Meeting & Scientific Sessions
Wednesday, 22 April – Saturday, 25 April, 2026
Metro Toronto Convention Centre
Toronto, ON Canada
Featured Poster Presentations

Add non-CME Featured Poster Sessions to your ISHLT2026 agenda. Held in a theater located within the Poster Hall during the evening poster sessions, this 60-minute, rapid-fire session will feature five presentations selected from high-scoring poster abstracts focused in basic science and other topics. Presenters will have the unique opportunity to deliver a brief oral presentation of their abstract, rather than a traditional poster format.
CME is not offered for poster sessions.
Featured Poster Session 01
Wednesday, 22 April | 6:00 – 7:00 p.m.
Basic Science - Predictive Molecular Signatures
Poster Theater 1
Deep Cardiovascular Phenotyping Reveals Specific Hemodynamic, Structural and Molecular Alterations and Compartmental RNA Signatures in Right Ventricular Pressure Overload Heart Failure
427 – Deep Cardiovascular Phenotyping Reveals Specific Hemodynamic, Structural and Molecular Alterations and Compartmental RNA Signatures in Right Ventricular Pressure Overload Heart Failure
Featured Poster Session 1 (Non-CME): Basic Science – Predictive Molecular Signatures
Wednesday, 22 April | 6:00 –
7:00 p.m. | Poster Theater 1

G. Hansmann1, E. Legchenko2, D. Bobylev3, J. Sorensen Axelsen4, K. Hysko2, H. Hasan2, A. Andersen4, S. Andersen4, E. Rog-Zielinska5, M. Meier6, A. Klinke7, P. Chouvarine2. 1Department of Anesthesiology and Intensive Care Medicine, Eberhard Karls University Tuebingen, Tuebingen, Germany, 2Department of Pediatric Cardiology and Critical Care, Hannover Medical School, Hannover, Germany, 3Cardiothoracic, Transplantation and Vascular Surgery, Hannover Medical School, Hannover, Germany, 4Department of Cardiology, Aarhus University Hospital, Aarhus, Denmark, 5Institute for Experimental Cardiovascular Medicine, University Heart Center and Faculty of Medicine, University of Freiburg, Freiburg, Germany, 6Small Animal Imaging Centre, Central Animal Facility, Hannover Medical School, Hannover, Germany, 7Dept of General and Interventional Cardiology/Angiology, Heart and Diabetes Ctr N Rhine-Westphalia, University Hospital of the Ruhr-Universität Bochum, Bad Oeynhausen, Germany.
Abstract
Purpose
We have identified RNA regulatory networks specific to pressure afterload-induced RV hypertrophy in human non-failing congenital heart disease (tetralogy of Fallot). Here, we explore hemodynamics, cardiomyofiber/-cyte structure and function, and compartmental regulatory RNA networks in RV pressure overload heart failure in pulmonary artery-banded (PAB) rats.
Methods
4-5 weeks old male SD rats underwent surgical clipping of the PA trunk (PAB; n=8), or Sham procedure (n=7). 4 weeks after surgery, spontaneously breathing rats underwent echocardiography, cardiac MRI/1H-MRS and closed-chest right-left-heart catheterization, followed by ex-vivo MR high resolution diffusion tensor imaging (MRhrDTI), histology/IF, EM, and ventricle-specific RNA-Seq analysis of competing endogenous (circ, lnc) RNA, mi- and mRNAs.
Results
PAB vs. Sham rats had greatly elevated RVSP (83 vs. 27 mmHg; p<0.0001) and RVEDP (11 vs. 5 mmHg; p<0.001), while LV and aortic pressures were similar. Non-invasive imaging showed RV systolic and diastolic dysfunction, hypertrophy and dilation, and lower creatinine and fatty acid signals, in PAB vs. Sham RVs. In MRhrDTI, packing and integrity of myocytes in the RV was impaired (MD map), and RV fiber orientation vs. epicardial plane was decreased (lower helix angle gradient). We found RV fibrosis (strongest in ventricular-septum hinge points), capillary rarefaction and cardiomyocyte hypertrophy, along with a reduction of mitochondrial size and an increased percentage of damaged mitochondria. RNA-Seq unraveled boosted cardiac pathways associated with apoptosis, DNA damage, angiogenesis, reduced fatty acid oxidation, increased glycolysis, hypoxia, reactive oxygen species, inflammation, and fibrosis. The strongest pro-fibrotic and heart failure-associated miR-regulation occurred in the septal hinge points (pro-fibrotic: miR-132, miR-21, miR-34a; heart failure: miR-18a, miR-221, miR-223, miR-652), followed by changes in the RV anterior and LV posterior walls.
Conclusion
Septal hinge point-enhanced fibrosis, altered RV fiber orientation, cardiomyocyte mitochondrial damage, lower creatinine and fatty acid signals, in conjunction with complex, compartmental regulatory RNA networks, characterize RV pressure overload heart failure.
Donor-Recipient Sex Mismatch and Circulating dd-cfDNA Drive Molecular Rejection Signatures After Heart Transplantation
428 – Donor-Recipient Sex Mismatch and Circulating dd-cfDNA Drive Molecular Rejection Signatures After Heart Transplantation
Featured Poster Session 1 (Non-CME): Basic Science – Predictive Molecular Signatures
Wednesday, 22 April | 6:00 –
7:00 p.m. | Poster Theater 1

A. Fernandez Valledor1, A. Hertz1, C. Moeller1, J. Baranowska1, D. Oren1, S. Rahman1, C. Hennecken1, I. Richter1, A. Rahman1, B. Elad1, D. Lotan1, E. M. DeFilippis1, A. Yunis1, J. Fried1, J. Raihkelkar1, K. T. Oh1, D. Bae1, M. Regan2, D. T. Majure2, F. Latif1, K. D. Clerkin1, N. Urielv, G. Sayer1. 1Columbia University Irving Medical Center, New York, NY, 2NewYork-Presbyterian Hospital, New York, NY.
Abstract
Purpose
Sex mismatch between donor and recipient has been associated with immune activation and worse outcomes after heart transplantation (HT), yet its relationship with molecular rejection pathways remains unclear.
Methods
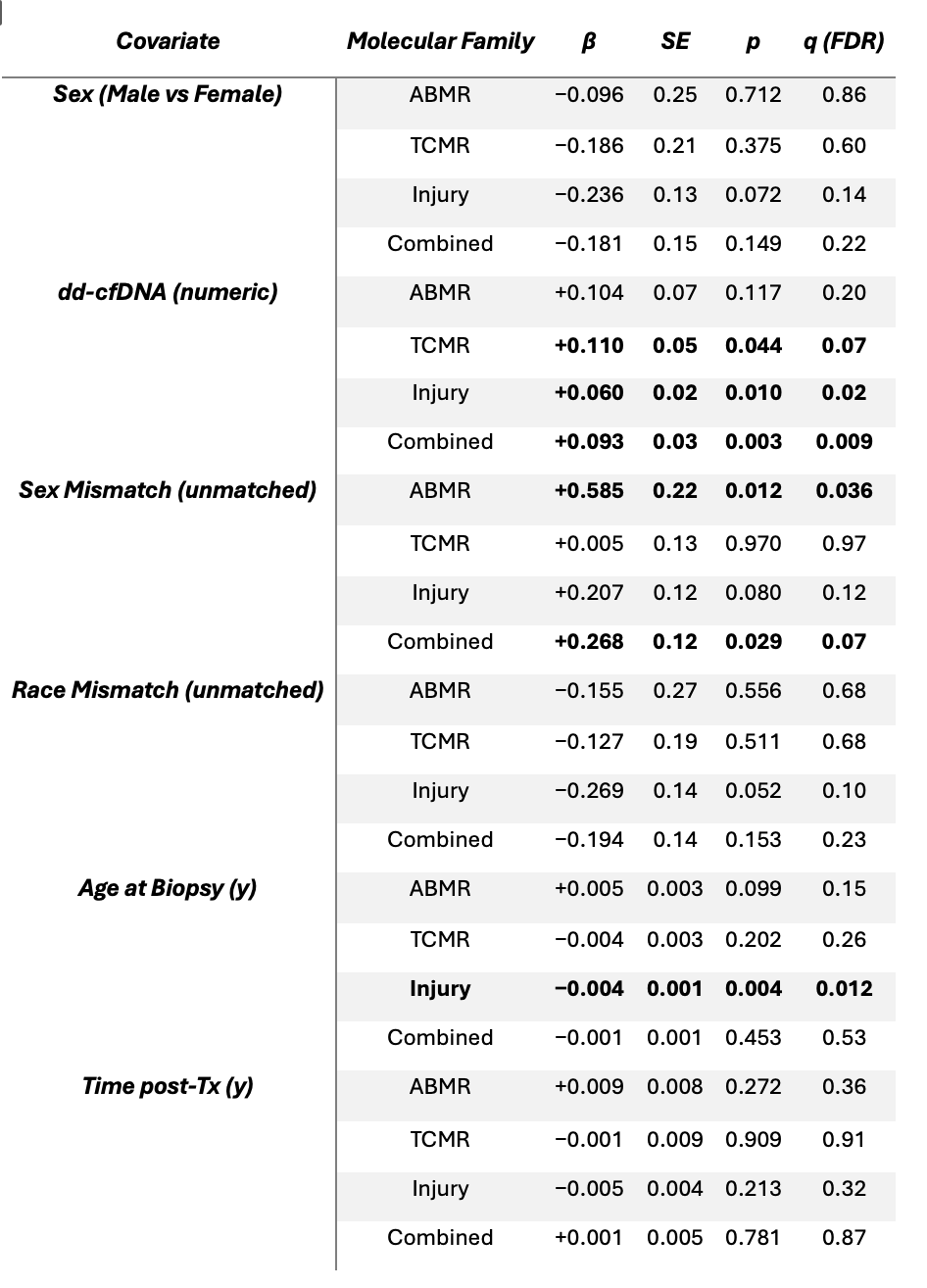
Endomyocardial biopsies from a prospective multicenter registry 11/2022-03/2025) were analyzed using the Molecular Microscope (MMDx). Transcript families representing antibody-mediated rejection (ABMR), T-cell-mediated rejection (TCMR), and injury were summarized as family-level z-scores and also combined to generate an overall molecular activation index. Associations between sex and molecular activation were evaluated using generalized estimating equations (GEE) clustered by patient. Models included sex (female reference) as the main exposure, adjusting for dd-cfDNA, donor-recipient sex and race mismatches, age at biopsy, and time since HT.
Results
A total of 579 biopsies were identified; 399 from 221 patients with complete donor information, molecular, and biomarker data were included. Donor-recipient sex mismatch was independently associated with higher ABMR transcript expression (β = +0.59, p = 0.012, q = 0.04) and greater global molecular activation (β = +0.27, p = 0.03, q = 0.07). dd-cfDNA correlated with elevated TCMR (β = +0.11, p = 0.044), injury (β = +0.06, p = 0.010), and global activation (β = +0.09, p = 0.003), all remaining significant after FDR correction. Recipient sex, race mismatch, age, and time from HT were not independently associated (p>0.05; Table).
Conclusion
Donor-recipient sex mismatch, rather than recipient sex, was independently associated with higher ABMR activity and overall molecular activation. In contrast, circulating dd-cfDNA reflected a more global pattern of transcriptomic injury across rejection pathways. Together, these results suggest that sex mismatch and tissue injury represent distinct yet complementary mechanisms driving immune activation beyond conventional histologic assessment.
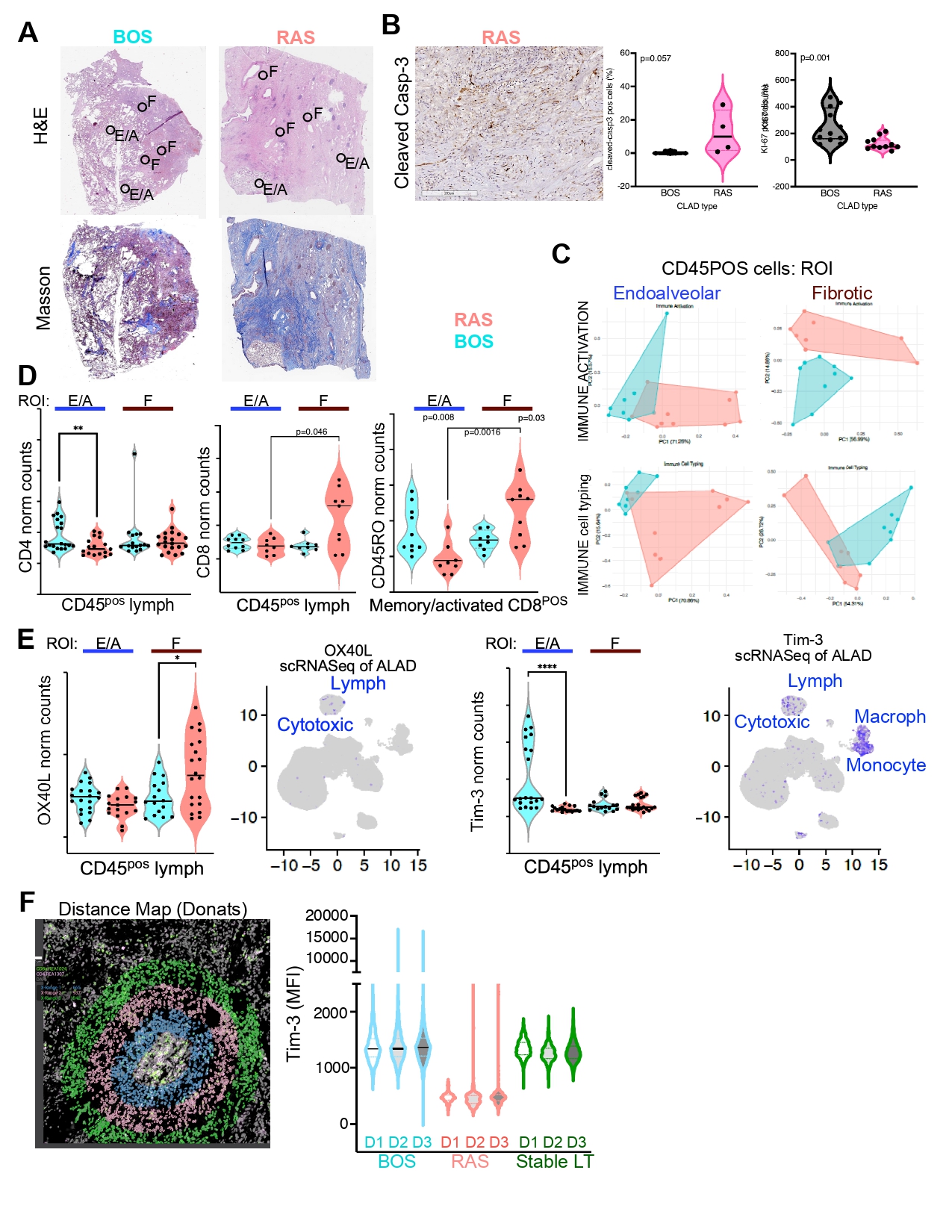
Transcriptomic Characteristics of Human Donor Lung Epithelial Cells at Single-Cell Resolution Are Related to Primary Graft Dysfunction
429 – Transcriptomic Characteristics of Human Donor Lung Epithelial Cells at Single-Cell Resolution Are Related to Primary Graft Dysfunction
Featured Poster Session 1 (Non-CME): Basic Science – Predictive Molecular Signatures
Wednesday, 22 April | 6:00 –
7:00 p.m. | Poster Theater 1

J. Li, S. Keshavjee, M. Liu. Toronto Lung Transplant Program and Latner Thoracic Research Laboratories, Toronto General Hospital, University of Toronto, Toronto, ON, Canada.
Abstract
Purpose
Primary Graft Dysfunction (PGD) contributes to early lung transplantation (LTx) morbidity and mortality. Donor lung quality affects the development of PGD, however, bulk transcriptomics has been challenging to identify PGD-specific genes. Single cell RNA sequencing (scRNA-seq) enables the capture of gene expression in a cell type specific manner. We examined epithelial transcriptomic profiles in donor lungs based on the severity of PGD.
Methods
scRNA-seq was performed on donor lung biopsies from 6 LTx cases. Cases were grouped according to 72h-post-transplant PGD grade (3 cases: 0-1; 3 cases: 2-3). Gene expression in different epithelial cells were compared between the two PGD groups. Gene Set Enrichment Analysis (GSEA) was used to identify differentially expressed Gene Ontology Biological Process (GOBP) pathways. Representative genes were selected.
Results
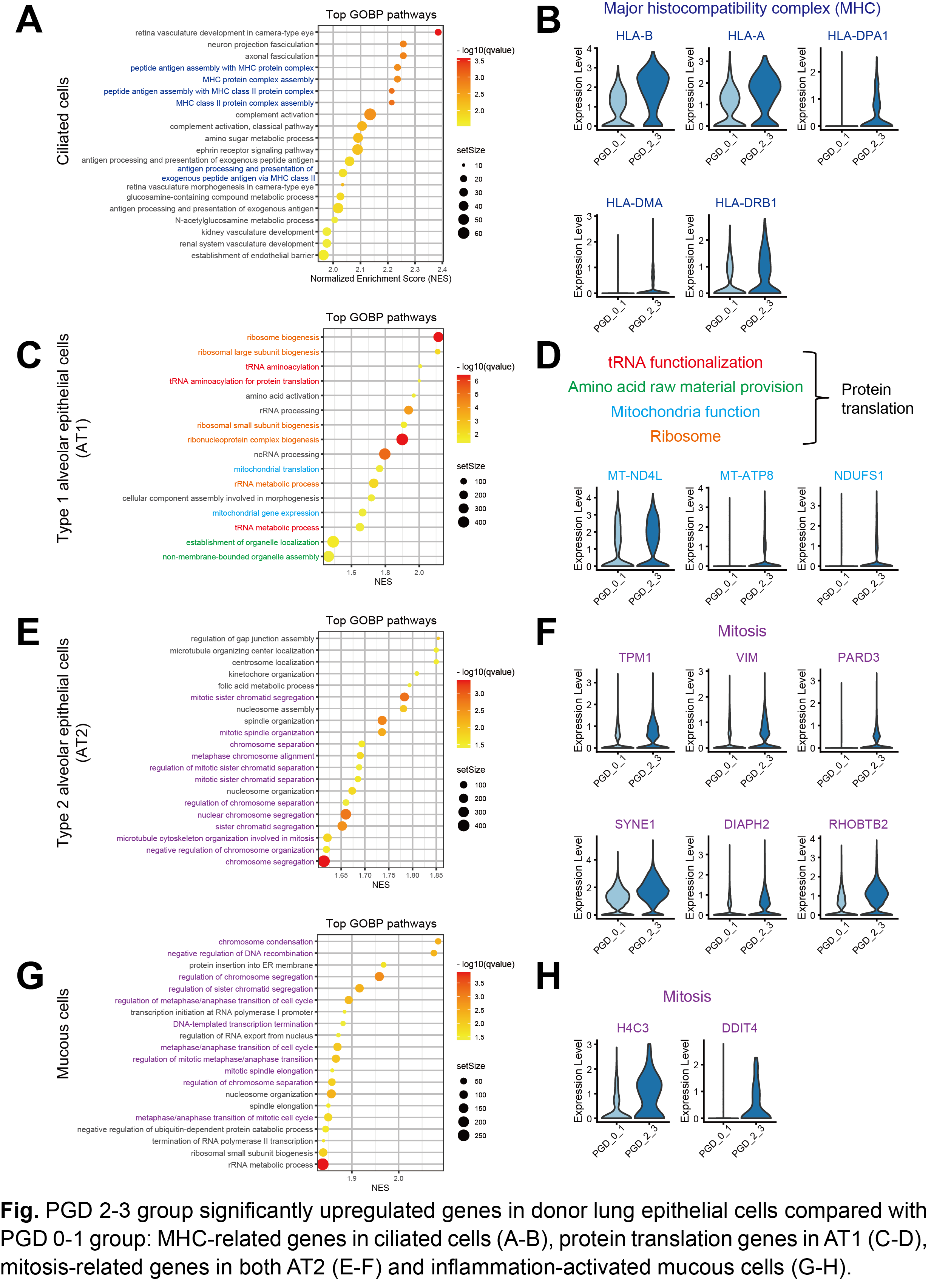
Compared with the PGD 0-1 group, the PGD 2-3 group showed distinct gene expression patterns in four epithelial cell types. In ciliated cells, MHC-related genes were notably upregulated (Fig. A, B). In type 1 alveolar epithelial cells (AT1), genes closely related to protein translation were significantly upregulated (Fig. C, D). Both type 2 alveolar epithelial cells (AT2, Fig. E, F) and mucous cells exhibited significantly higher expression of mitosis-related genes (Fig. G, H).
Conclusion
Using scRNA-seq, differentially expressed genes and pathways were found in donor lung epithelial cells. Gene expression in other cell types is further studied with scRNA-seq. Understanding cellular and molecular mechanisms in donor lungs may improve our donor lung selection, preservation and treatment to prevent PGD.
Single Cell Transcriptomic Landscape of Dilated Cardiomyopathy Across the Age-Spectrum Reveals Amplified Pediatric Response to Mechanical Stress
430 – Single Cell Transcriptomic Landscape of Dilated Cardiomyopathy Across the Age-Spectrum Reveals Amplified Pediatric Response to Mechanical Stress
Featured Poster Session 1 (Non-CME): Basic Science – Predictive Molecular Signatures
Wednesday, 22 April | 6:00 –
7:00 p.m. | Poster Theater 1

H. J. Tadros1, D. Turaga1, C. Tsai2, Y. Zhao3, K. Hope1, L. Wadhwa4, I. Adachi4, X. Li3, J. F. Martin2. 1Pediatrics, Texas Children’s Hospital/ Baylor College of Medicine, Houston, TX, 2Department of Integrative Physiology, Baylor College of Medicine, Houston, TX, 3The Texas Heart Institute at Baylor College of Medicine, Houston, TX, 4Department of Surgery, Texas Children's Hospital/ Baylor College of Medicine, Houston, TX.
Abstract
Purpose
Pediatric dilated cardiomyopathy (pDCM) is the leading cause of heart failure in children and carries a high risk of mortality. pDCM remains understudied compared to its adult (aDCM) counterpart. We hypothesize that pDCM will exhibit unique transcriptomics compared to aDCM.
Methods
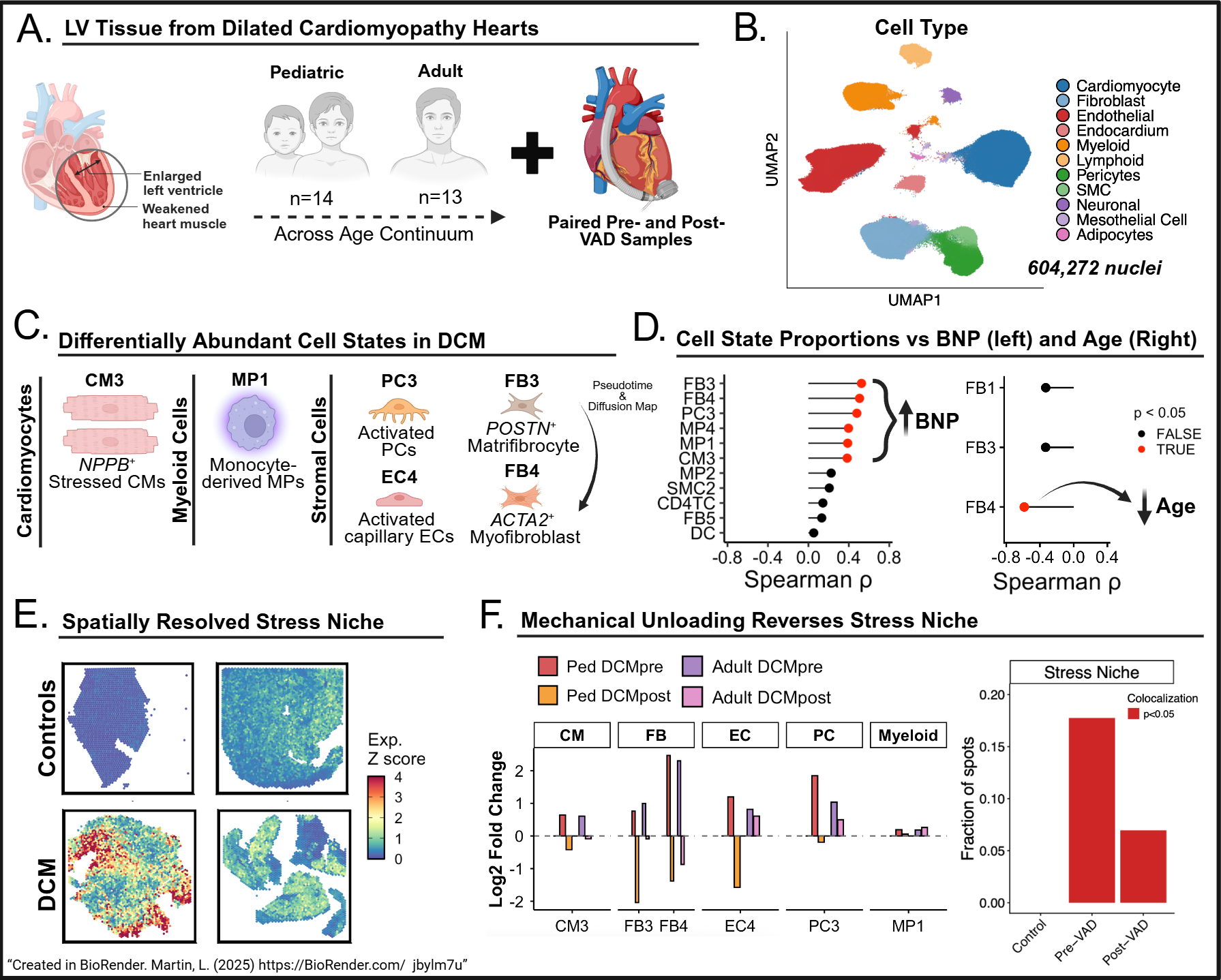
We used the 10x Genomics platform to perform single-nucleus RNA sequencing (snRNA-seq) on paired left ventricle pDCM samples (n=28) before and after ventricular assist device (VAD) placement. We integrated data from pediatric (n=8) and adult (n=14) controls, and paired aDCM samples (n=26) to compare differences (Fig 1A). We utilized the spatial transcriptomic platform, Visium, to analyze pediatric donors (n=2) and pDCM pre-VAD (n=2) and post-VAD (n=2).
Results
We integrated 604,272 nuclei from 76 samples. Unbiased clustering revealed 11 cardiac cell types and 35 cell states (Fig 1B). We identified cell states enriched in DCM (Fig 1C), including two fibroblast populations termed FB3 (POSTN+ matrifibrocytes) and FB4 (ACTA2+ myofibroblasts). FB3 appears to gives rise to FB4, with increasing signs of mechanical stress reflected by progressive expression of Yap target genes and mechanosensitive channels (PIEZO1, PIEZO2). FB4 showed age-dependent enrichment in pDCM (Fig 1D). Similarly, Yap target genes were enriched in NPPB+ cardiomyocyte cluster CM3 and pathologic cell states directly correlated with BNP (Fig 1D). Co-localization was exhibited on spatial transcriptomics, suggesting a spatially resolved niche (Fig 1E). Following VAD unloading, there was an amplified decrease in pathologic cell states in pDCM compared to aDCM and less spatial colocalization (Fig 1F).
Conclusion
This is the largest pediatric DCM snRNA-seq dataset and the first to profile DCM across ages, revealing age-dependent fibroblast mechanosensing and a stressed, spatially resolved niche more effectively rescued by VAD in pDCM. We also highlight future directions for research into pDCM.
Early Transcriptomic Injury After Heart Transplantation Predicts Adverse Outcomes
431 – Early Transcriptomic Injury After Heart Transplantation Predicts Adverse Outcomes
Featured Poster Session 1 (Non-CME): Basic Science – Predictive Molecular Signatures
Wednesday, 22 April | 6:00 –
7:00 p.m. | Poster Theater 1

A. Fernandez Valledor1, C. Moeller1, A. Hertz1, J. Baranowska1, D. Oren1, S. Rahman1, C. Hennecken1, I. Richter1, A. Rahman1, B. Elad1, D. Lotan1, E. M. DeFilippis1, A. Yunis1, J. Fried1, J. Raihkelkar1, K. T. Oh1, D. Bae1, M. Regan2, D. T. Majure2, F. Latif1, K. D. Clerkin1, N. Uriel1, G. Sayer1. 1Columbia University Irving Medical Center, New York, NY, 2NewYork-Presbyterian Hospital, New York, NY.
Abstract
Purpose
Despite advances in perioperative management, ischemia-reperfusion injury remains common after heart transplantation (HT). Transcriptomic profiling enables objective quantification of early graft injury, but its prognostic significance is uncertain. We evaluated whether parenchymal injury detected by the Molecular Microscope (MMDx) predicts short-term adverse outcomes after HT.
Methods
Biopsies obtained within 56 days post-HT from a prospective multicenter registry (2022-2025) were analyzed. A composite MMDx injury z-score was derived from predefined transcripts (HT1, IRRAT, S4, QCMAT) and assessed continuously (per 1-SD) and by quartiles (Q4 vs Q1-Q3). Outcomes at 90, 180, and 365 days included death, graft dysfunction (LVEF <50%), histologic or molecular rejection, and worsening donor-derived cell-free DNA (dd-cfDNA). Logistic and mixed-effects models were clustered by patient and adjusted for age, sex, and time from HT.
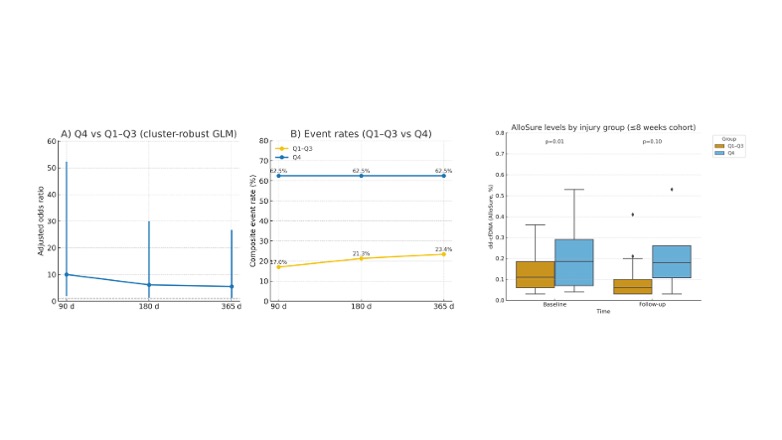
Results
Among 63 early biopsies, composite adverse events occurred in 18, 20, and 20 cases at 90, 180, and 365 days. Event rates increased stepwise across injury quartiles (12-25% in Q1-Q3 vs 63% in Q4), reflecting a 2-5-fold higher risk in Q4. High injury (Q4 vs Q1-Q3) predicted events at 90 days (OR 10.0, 95% CI 1.9-52.3; p=0.006) and remained significant at 180 and 365 days (OR 6.1, 95% CI 1.3-29.9; p=0.025). The continuous score also predicted 90-day events (OR 2.5, 95% CI 1.0-6.2; p=0.041). Events in Q4 were mainly due to molecular rejection (44%) and dd-cfDNA elevation (38%). dd-cfDNA was higher in Q4 at baseline (0.185% vs 0.110%; p=0.01) and follow-up (median 42 days), while it declined in Q1-Q3 (to 0.055%). In mixed models, Q4 biopsies showed persistently elevated dd-cfDNA (β=+0.09%, 95% CI 0.02-0.16; p=0.01).
Conclusion
Early transcriptomic injury identifies patients at higher risk of molecular activation and adverse outcomes. The initial ischemic and inflammatory insult may set the stage for ongoing antigenic exposure, fueling immune activation and persistent graft injury.
Nursing & Allied Health
Poster Theater 2
Consensus Conference on the Evolving Role of Nursing Professionals in Thoracic Transplantation
432 – Consensus Conference on the Evolving Role of Nursing Professionals in Thoracic Transplantation
Featured Poster Session 1 (Non-CME): Nursing & Allied Health
Wednesday, 22 April | 6:00 –
7:00 p.m. | Poster Theater 2

B. Coleman1, A. Velleca2, C. Sumbi3, C. Benden4, A. Guzman1, J. Kobashigawa3. 1Cedars-Sinai Medical Center, Los Angeles, CA, 2Cedars-Sinai Smidt Heart Inst & Comprehensive Transplant Ctr, Los Angeles, CA, 3Cedars-Sinai Smidt Heart Institute, Los Angeles, CA, 4Boston Children's Hospital, Boston, MA.
Abstract
Purpose
Since the first ISHLT Thoracic Nursing Consensus Conference in 2015, the complexity of cardiothoracic transplantation along with administrative demands, workload intensity, and need for rapid, precise communication among multidisciplinary teams continues to place significant pressure on transplant coordinators (TCs) and advanced practice providers (APPs). Rapid adoption of emerging technologies such as organ care systems, ex vivo lung perfusion (EVLP), normothermic regional perfusion (NRP), and other advanced preservation platforms has expanded transplantation, often outpacing workforce capacity and training. We convened the 2nd ISHLT Thoracic Nursing Consensus Conference to assess how evolving demands affect professional development/sustainability of TC and APP roles worldwide.
Methods
Using purposive sampling, 75 nurses representing 8 countries participated in an international consensus forum. Two additional focus groups (n=27) were held at times accessible to global participants (Table 1). Conference attendees engaged in didactic sessions and structured breakout discussions facilitated by moderators. Group findings were synthesized and refined in plenary sessions, with final content compiled by consensus leads for analysis.
Results
Thematic analysis identified 7 domains: (1) staffing models and workload; (2) work environment, resilience, and retention; (3) roles and qualifications; (4) professional development and nursing value; (5) clinical practice processes; (6) teaching and innovation; (7) patient education and access. 13 actionable recommendations emerged to strengthen role sustainability and system performance.
Conclusion
While transformative technologies enhance survival and access to thoracic transplantation, they also intensify workforce strain. Consensus participants emphasized strategic investments in staffing, recognition, and development pathways to sustain the vital contributions of TCs and APPs to advanced cardiothoracic care.
Genetic Testing is Indicated for All Patients with Non-Ischemic End Stage Cardiomyopathy
433 – Genetic Testing is Indicated for All Patients with Non-Ischemic End Stage Cardiomyopathy
Featured Poster Session 1 (Non-CME): Nursing & Allied Health
Wednesday, 22 April | 6:00 –
7:00 p.m. | Poster Theater 2

C. Pine1, J. Herre2, A. Harper2, A. Toepp3. 1Advanced Heart Failure, Sentara Healthcare, Norfolk, VA, 2Advanced Heart Failure, Sentara Norfolk General Hospital, Norfolk, VA, 3Sentara Norfolk General Hospital, Norfolk, VA.
Abstract
Purpose
Genetic abnormalities are found in 5 to >20% of cases of dilated cardiomyopathy (DCM), depending on the population studied. Truncating variants of TTN (TTNtv) are the most common genetic cause of DCM, accounting for up to 25% of cases with familial inheritance. Lifetime risk for DCM is high but recent studies have shown that treatment of TTNtv pre-DCM may delay development of symptoms.
Methods
We studied 198 patients (pts) with non-ischemic DCM and end stage heart failure defined by need for heart transplantation or durable mechanical support (Tx/VAD). Presumed etiology was malignancy (1), congenital (2), anthracycline (4), myocarditis (2), peripartum (2), hypertrophic (4), valvular (1), infiltrative (2), mixed ischemic/nonischemic (2) and familial (14). The remaining pts had no etiology identified.
Results
Pts were 65% male; mean age 51 years; 56% were Black and 39% were White. Initial treatment was VAD in 32 pts and tx in166 pts. Pts were studied using the Invitae/Labcorp cardiomyopathy panel (121 genes) in 164 pts, the Ambry panel (92 genes) in 33 pts and Boston University genetic testing in 1 pt. Pathogenic or potentially pathogenic variants were noted in 67/198 (33.8%) of patients. Variants of the TTN gene were seen in 33/198 (16.7%) pts. No single variant appeared in more than 1 pt. TTR variants (all Val142Ile) were seen in 9/198pts (5.6%). Among pts with a family history of DCM, 10/14 had variants of genes associated with DCM. Among the 184 patients with no family history of DCM, 33 had TTNtv. Other pathogenic variants included ACTC1 (1), AGL (2), BAG3 (1), CPT2 (1), DMD (2), DNAJC19 (1), FKTN (1), FLNC (3), LMNA (3), LZTR1 (2), MTO1 (1), MYBPC3 (3), MYH7(4), PCCA (1), PCCB (1), PKP2 (2), PLN (1), PRKAG2 (1), SDHA (1), SLC22A5 (1), TCAP (1), TNNI3 (1), CAV3 (1), and TRDN (1). Only 3/33 patients with TTNtv had family histories of DCM. Among patients with pathogenic or potentially pathogenic variants, 237 siblings and 152 children were identified. Among the 62 family members tested, 36 had shared genes.
Conclusion
1. Genetic abnormalities are common in patients with end-stage cardiomyopathy even in the absence of a family history. 2. Variants of the TTN gene are the most common genetic cause for nonischemic cardiomyopathy, occurring in 16.7% of patients with DCM undergoing Tx/VAD. 3. Genetic testing will identify family members of TTNtv patients and allow early potentially life-saving treatment.
Exploring Motivation to Become a Peer Mentor Among Heart Transplant Recipients
434 – Exploring Motivation to Become a Peer Mentor Among Heart Transplant Recipients
Featured Poster Session 1 (Non-CME): Nursing & Allied Health
Wednesday, 22 April | 6:00 –
7:00 p.m. | Poster Theater 2

A. Assabiny1, O. Papp-Zipernovszky2, B. Ehrenberger2, J. Otohal2, S. Kugler3, A. Nagy2, I. Hartyánszky2, B. Sax2, B. Merkely2, B. Dávid4, Z. Ocsovszky2. 1Semmelweis University Heart and Vascular Center, Budapest, Hungary, 2Semmelweis University Heart and Vascular Centre, Budapest, Hungary, 3Semmelweis University, Heart and Vascular Center, Budapest, Hungary, 4Institute of Mental Health, Semmelweis University Institute of Mental Health, Budapest, Hungary.
Abstract
Purpose
During heart transplantation (HTX), peer support can help patients better understanding of their treatment and maintain mental well-being. We aimed to explore the factors underlying the motivation to become a peer mentor among heart transplant recipients.
Methods
We included HTX patients under follow-up at our clinic to fulfill a questionnaire. Motivation to become a mentor was assessed using an 11-point Likert scale (0-10). We also evaluated the quality of peer support across four stages of patient journey: before waiting list (preWL), during the waiting list (WL), intensive care unit (ICU), first posttransplant year (FY). Quality of peer support was classified as: (1) “highly helpful” (2) “neutral/slightly/moderately helpful” (3) “did not receive, but would have requested” and (4) “did not receive, did not requested”. Psychosocial status was assessed by employment status, quality of life (QoL, EQ-5D visual analogue scale), perceived social support (MSPSS), and depressive symptoms (BDI-9).
Results
201 recipients were included in the study (75.6% male, mean age 56.3 ± 11.5 years, 6.4 ± 0.3 years after transplant). The mean motivation to become a mentor was 8.2 ± 2.7, with 111 patients selecting the maximum score (10). Motivation correlated significantly with perceived support from friends (r(181) = 0.177; p = 0.016), QoL (r(187) = 0.247; p < 0.001), and negatively with depression scores (r(179) = −0.212; p = 0.004). Employed patients showed higher motivation (t(185) = −2.474; p = 0.014). Motivation was significantly higher among those who experienced peer support as “highly helpful” preWL as compared with all other subgroups (F(4) = 3.450; p = 0.0018). In a multivariate regression model motivation was predicted significantly by the following independent variables: peer support across the four stages, the QoL and BDI-9 (F(6) = 5.029; p < 0.0001; R² = 0.155). Independent predictors were peer support during the preWL and WL periods.
Conclusion
Motivation to become a peer mentor is high among heart transplant recipients. Better QoL and fewer depressive symptoms are associated with greater motivation. Experiences of peer support before transplantation appear to play a key role in shaping motivation for mentorship. Understanding these factors may support future peer support interventions.
Structured Ward-Based Assessment for Safe Transfer of ICU Patients Supported with Temporary Ventricular Assist Devices: A Single-Center Experience
435 – Structured Ward-Based Assessment for Safe Transfer of ICU Patients Supported with Temporary Ventricular Assist Devices: A Single-Center Experience
Featured Poster Session 1 (Non-CME): Nursing & Allied Health
Wednesday, 22 April | 6:00 –
7:00 p.m. | Poster Theater 2

H. Lin, Y. Yung, H. Liu, C. Tsao, Y. Chen, N. Chi. Nursing, National Taiwan University Hospital, Taipei, Taiwan.
Abstract
Purpose
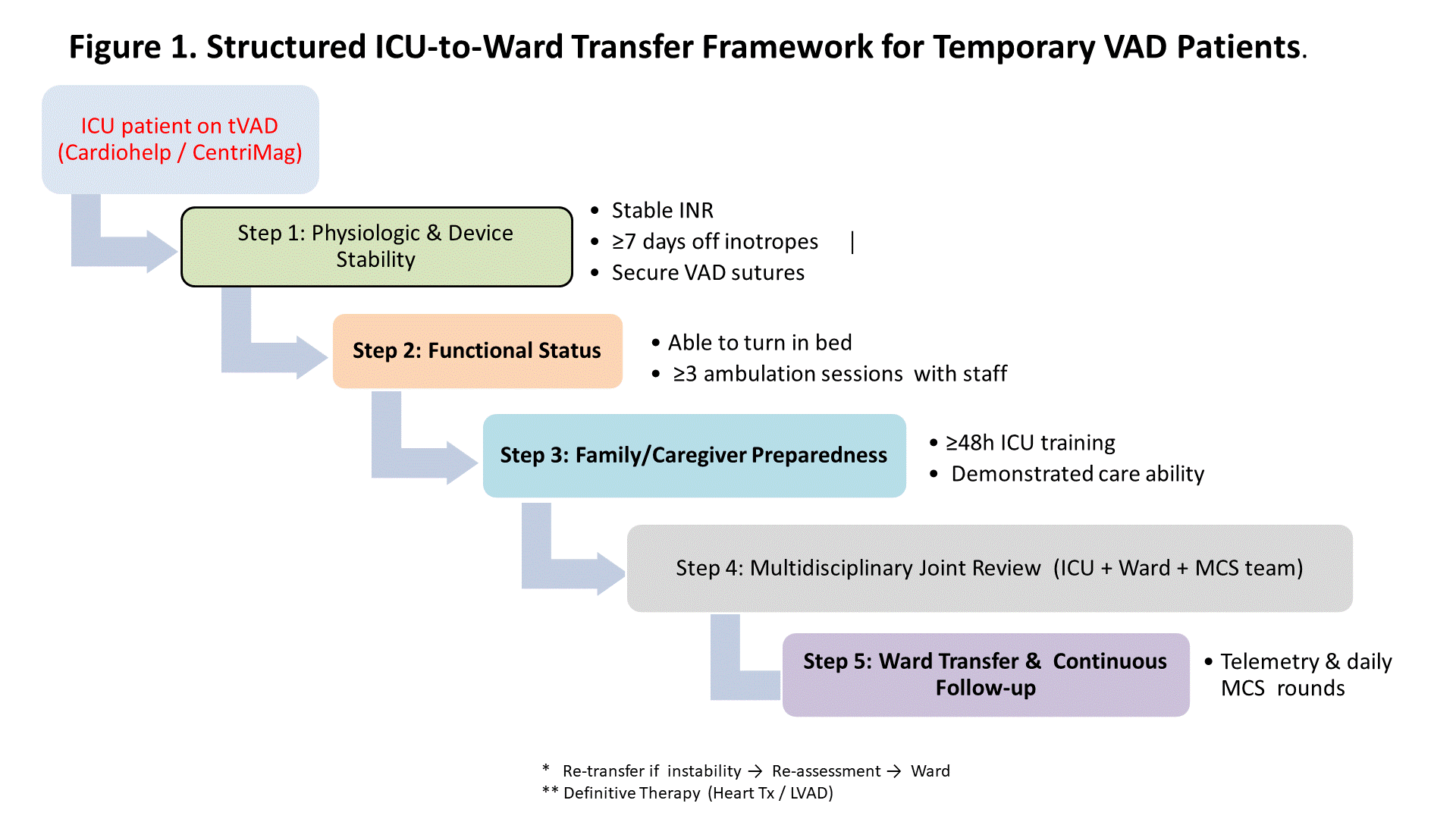
Temporary ventricular assist device (tVAD) support is typically initiated in the ICU for patients with acute advanced heart failure. Transitioning these patients to the general ward can optimize ICU resource use but raises safety and workload concerns. Our center developed a structured multidisciplinary evaluation system. This study assessed its safety, feasibility, and effectiveness in guiding centrifugal tVAD patients from ICU to ward while ensuring stable progression to definitive therapy.
Methods
From January 2014 to December 2024, 36 patients supported with centrifugal tVADs (Cardiohelp or CentriMag) were evaluated for ICU-to-ward transfer. Readiness was assessed in three domains: (1) Physiologic & Device—stable INR, ≥7 days off inotropes, secure driveline; (2) Functional—ability to turn in bed, ≥3 ambulation sessions with staff; (3) Family/Caregiver—≥48h ICU training, demonstrated caregiving ability. Transfer and re-transfer decisions were made jointly by ICU physicians, ward nurses, and the MCS team (see Fig1).
Results
All 36 patients (100%) were successfully transferred after a median ICU stay of 44.5 days (IQR 21.5-62.3). Median ward stay before definitive therapy was 36.5 days (IQR 17.3-59). Eight patients (22%) required ICU readmission—seven “two-in-two-out”, one “three-in-three-out”—due to transient hemodynamic or bleeding issues. All patients completed definitive therapy: heart transplantation (n=33, 91.7%) or durable LVAD (n=3, 8.3%). No mortality or major device-related events occurred during ward care.
Conclusion
A structured, multidisciplinary ICU-to-ward framework integrating physiologic, functional, and caregiver readiness domains enabled safe, resource-efficient ward management for tVAD patients. Even with intermittent ICU readmissions, this approach supported stable recovery and seamless progression to definitive therapy.
EVLP: Nurse-Led Innovation in Transplant Research and Career Advancement
436 – EVLP: Nurse-Led Innovation in Transplant Research and Career Advancement
Featured Poster Session 1 (Non-CME): Nursing & Allied Health
Wednesday, 22 April | 6:00 –
7:00 p.m. | Poster Theater 2

A. Ninan1, S. Houser1, D. Emmerton2, M. Blake2, S. Bollineni3, J. Murala3. 1UTSW, Dallas, TX, 2UT Southwestern Medical Center, Dallas, TX, 3UT Southwestern, Dallas, TX.
Abstract
Purpose
Ex vivo perfusion technologies have become an essential component of thoracic organ transplantation worldwide, creating a growing need for trained personnel to manage these devices. This project highlights the creation and impact of a nurse-led Ex Vivo Lung Perfusion (EVLP) program at our transplant center. As one of the few centers performing EVLP without perfusionist involvement, this model demonstrates how transplant nurses have advanced their clinical expertise, research engagement, and career growth through leadership in complex perfusion procedures.
Methods
The EVLP program was established in 2015 as a nurse-driven model in collaboration with transplant surgeons, pulmonologists, and research staff. Nurses received specialized training in circuit setup, perfusion management, and system troubleshooting, allowing independent operation of the EVLP platform. Most nurses had cardiovascular ICU experience and prior exposure to ECMO and ventricular assist devices. Program data and outcomes were reviewed to evaluate performance and nursing advancement.
Results
Since inception, the team has participated in nearly 20 research projects and 50 clinical EVLP runs, with 34 lungs (68%) successfully transplanted. The group also contributed to eight preclinical porcine EVLP studies focused on metabolomics and continues to expand human lung trials. The nurse-led team consistently achieved safe and effective perfusion without perfusionist or intensivist support. Participation fostered significant professional growth, with several nurses advancing into roles such as CRNAs and ACNPs.
Conclusion
With evolving ex vivo technologies, trained CVICU transplant nurses are a vital, sustainable resource for EVLP programs. Managing these systems is a natural progression for nurses skilled in cardiac device support. We believe this is a financially affordable and sustainable model and demonstrates resource optimization for any healthcare system.
Featured Poster Session 02
Thursday, 23 April | 4:30 – 5:30 p.m.
Basic Science - Perfusion-Associated Gene Changes
Poster Theater 1
Combined Cardiorenal Perfusion During Prolonged Ex-Situ Heart Perfusion Improves Cardiac Function
437 – Combined Cardiorenal Perfusion During Prolonged Ex-Situ Heart Perfusion Improves Cardiac Function
Featured Poster Session 2 (Non-CME): Basic Science – Perfusion-Associated Gene Changes
Thursday, 23 April | 4:30 – 5:30 p.m. | Poster Theater 1

H. Krifi1, A. Alber1, A. Akamkam1, J. Hadchouel2, P. Galichon2, J. Blonde3, S. Rozencwajg4, J. Guihaire5. 1Cardiac Surgery and Transplantation, Marie Lannelongue Hospital, Le Plessis Robinson, France, 2CoRaKiD - Inserm UMR_S1155, Hôpital Tenon, Sorbonne Université, Paris, France, 3EDEN Tech, Paris, France, 4Anesthesiology, Marie Lannelongue Hospital, University of Paris Saclay, Le Plessis Robinson, France, 5Cardiac Surgery and Transplantation, Marie Lannelongue Hospital, University of Paris Saclay, Le Plessis-Robinson, France.
Abstract
Purpose
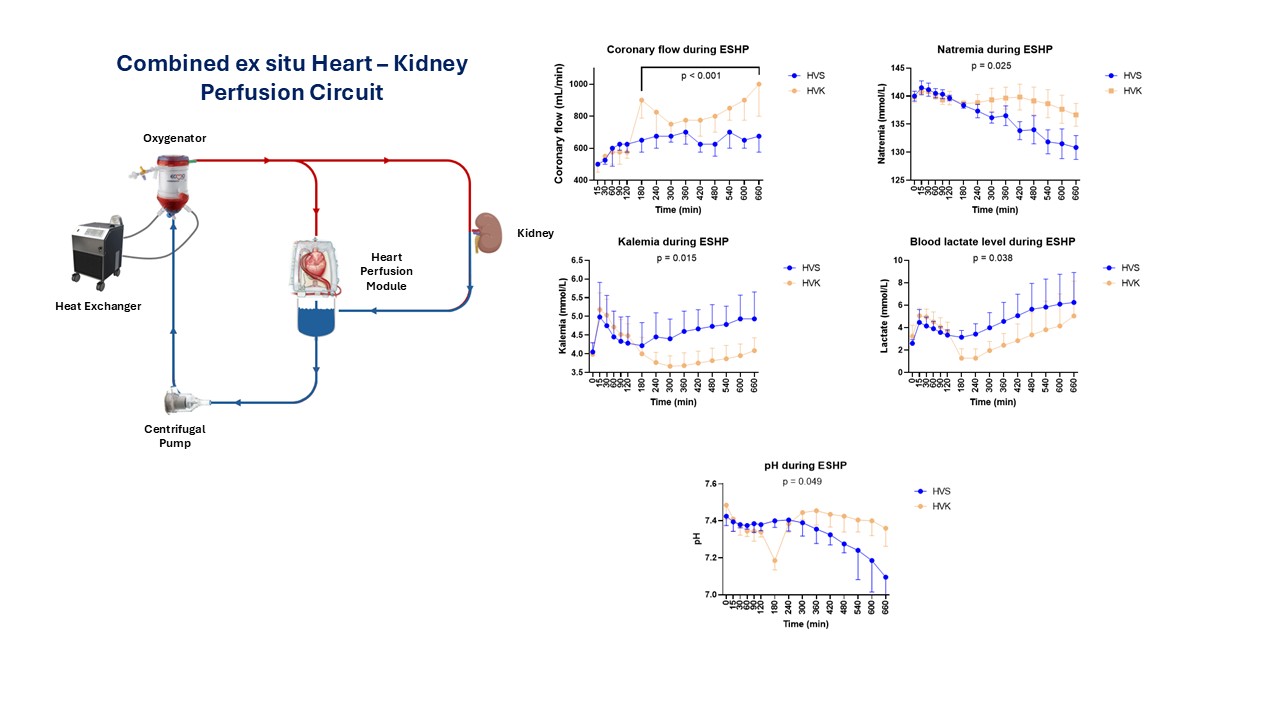
Normothermic ex-situ heart perfusion (ESHP) has been validated for isolated preservation of the donor heart. Concomitant perfusion of multiple organs has been poorly investigated. We hypothesize that combined cardiorenal perfusion for 10 hours allows for better preservation of the heart.
Methods
Porcine hearts were perfused with oxygenated blood in Langendorff mode for 12 hours combined with a 10-hour ex situ kidney perfusion (HVK group, n=6). This model was compared to an isolated ESHP model (HVS group, n=6). The kidney was positioned as a side branch of the main circuit, and coronary flow was adjusted to maintain aortic root pressure between 50 and 60 mmHg. Renal flow was maintained between 300 and 500 mL/h and perfusate samples were collected hourly. After 11 hours of perfusion, the hearts were turned to working mode to allow functional assessment.
Results
Median coronary flow was higher in HVK group from 3 to 11 hours of ESHP (800 [767-871] vs 667 [629-671] mL/min, p<0.001). After 11 hours of ESHP, all HVK hearts successfully transitioned to working mode, two HVS hearts (33.3%) failed at generating a mean aortic pressure above 50 mmHg. The kidney remained functional, with an average oxygen consumption of 6.4 ± 1.2 mL/min and low renal vascular resistance, with a median of 0.11 [0.08-1.13]. The mean sodium concentration in the blood perfusate was higher in HVK group (139 ± 1 vs 137 ± 4 mmol/L, p=0.025), the mean potassium concentration was lower (4.0 [3.8-4.5] vs 4.6 [4.3-5.0] mmol/L, p=0.015). The mean lactate level was higher in HVS group (4.4 ± 1.1 vs 3.4 ± 1.3 mmol/L, p=0.038). The median pH of the perfusate was higher in HVK group (7.39 [7.34-7.41] vs 7.37 [7.29-7.39], p=0.049) (Figure).
Conclusion
Combined cardiorenal perfusion was associated with improved homeostasis of the perfusate. These findings highlight the importance of blood purification during prolonged normothermic ESHP.
Optical-Flow Analysis Demonstrates Preserved Contractility and In-Vivo Function After 24-Hour of Hypothermic Oxygenated Perfusion in Porcine Hearts
438 – Optical-Flow Analysis Demonstrates Preserved Contractility and In-Vivo Function After 24-Hour of Hypothermic Oxygenated Perfusion in Porcine Hearts
Featured Poster Session 2 (Non-CME): Basic Science – Perfusion-Associated Gene Changes
Thursday, 23 April | 4:30 – 5:30 p.m. | Poster Theater 1

N. Ott1, I. Mason1, W. Mitchell1, M. Moroi1, Y. Kosuri1, S. Faruqi2, C. Goergen2, C. Camillo1, C. Karcher1, D. Albino1, A. Adamo1, K. Takeda1, G. Ferrari1. 1Columbia University, New York, NY, 2Purdue University, West Lafayette, IN.
Abstract
Purpose
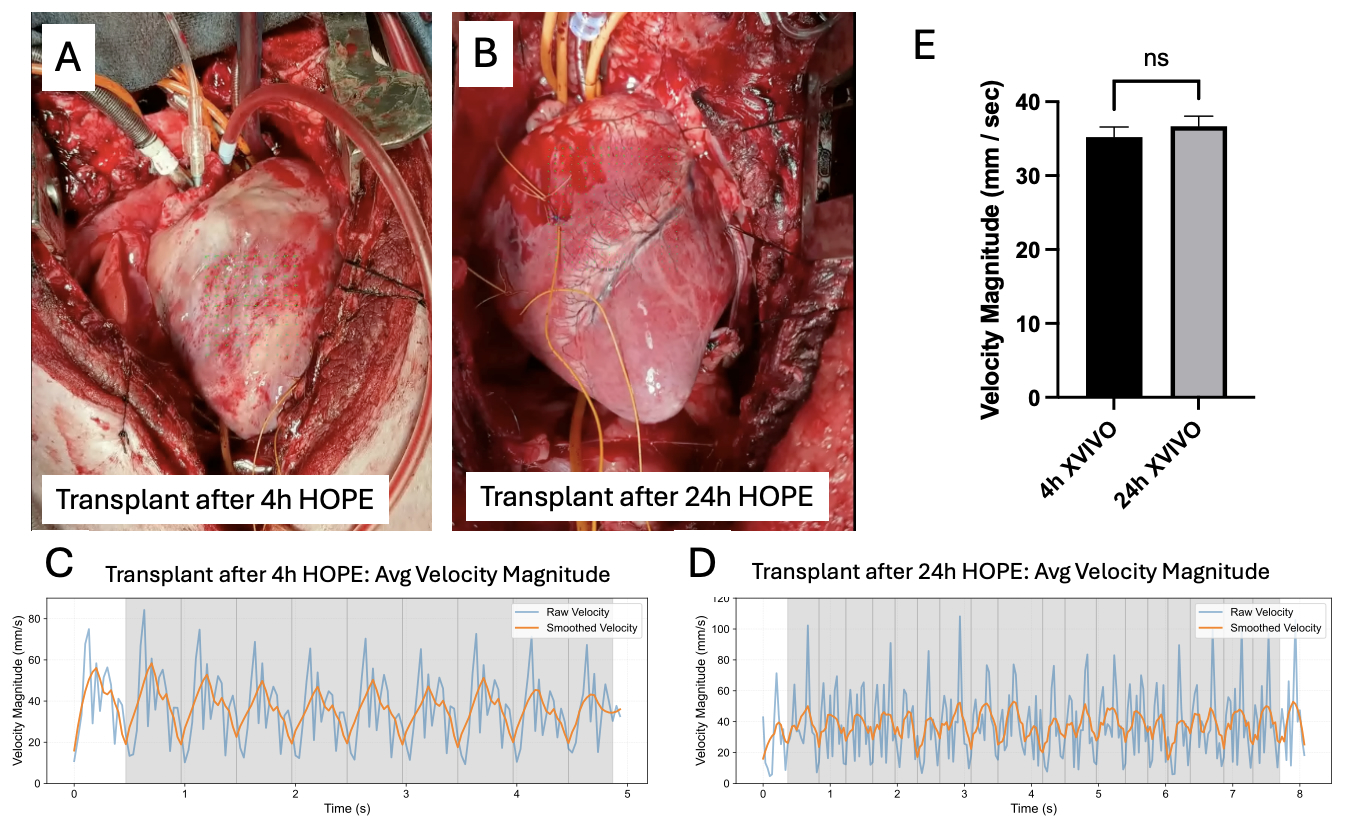
xVIVO’s Hypothermic Oxygenated Perfusion (HOPE) system extends cardiac allograft preservation beyond the 4-6 h limit of static cold storage (SCS). Here we assess sinus rhythm and coordinated contraction by optical-flow analysis to demonstrate functional recovery after 24h of hypothermic preservation and orthotopic transplantation (HTx) following HOPE in pigs.
Methods
Yorkshire pigs (n=11) underwent donor cardiectomy. Hearts were preserved by SCS (n=2) or HOPE (n=6; 3h, N=2; 24h, N=2, 48h, N=2) for up to 48h and reanimated for 2h using a bench-top normothermic machine perfusion (NMP). Histology, Cardiomyocyte integrity, transcriptomics and metabolomics were performed. 3 HOPE hearts (4h, N=1 and 24h, N=2) were transplanted into a recipient pig and contractile function was quantified using optical-flow motion analysis and LV pressure (dp/dt).
Results
HOPE preserved cardiomyocyte integrity and RNA quality through 48 h, while SCS showed loss of viability by 6 h. Transcriptomics revealed upregulation of mitochondrial and metabolic pathways and downregulation of apoptosis and inflammation in HOPE. All HOPE hearts regained sinus rhythm during NMP; the 24 h and 48 h groups showed contractile velocities comparable to 3 h controls, whereas 24 h SCS failed to reanimate. Optical-flow analysis demonstrated preserved average surface velocity (HOPE 4 h vs 24 h: p > 0.05). Orthotopic transplants after 4 h and 24 h HOPE reperfused successfully, recovering sinus rhythm and coordinated contraction in vivo.
Conclusion
HOPE maintains myocardial structure, molecular integrity, and contractile function for up to 48 h and supports successful orthotopic transplantation after 24 h preservation.
Cold Ischemia and Myocardial Function After Cardiac Autotransplantation - Establishment of a Novel Porcine Model
594 – Cold Ischemia and Myocardial Function After Cardiac Autotransplantation - Establishment of a Novel Porcine Model
Featured Poster Session 2 (Non-CME): Basic Science – Perfusion-Associated Gene Changes
Thursday, 23 April | 4:30 – 5:30 p.m. | Poster Theater 1

L. Micek1, J. Wollborn2, J. Scheumann3, S. Maier1, S. Neudorf1, M. Eschenhagen1, D. Damjanovic1, J. Dinkelaker4, H. Cristina Schmitz4, A. Dimov1, M. Kreibich1, T. Berger1, M. Czerny1, D. Schibilsky5. 1Department of Cardiovascular Surgery, University Hospital Freiburg Bad Krozingen, Freiburg, Germany, 2Department of Anaesthesiology, Brigham and Women's Hospital, Boston, MA, 3Department of Cardiology, University Hospital Freiburg Bad Krozingen, Freiburg, Germany, 4Center for Experimental Models and Transgenic Service, University Hospital Freiburg, Freiburg, Germany, 5Department of Cardiac Surgery, Lucerne Cantonal Hospital, Lucerne, Switzerland.
Abstract
Purpose
Cold ischemia remains a key determinant of graft function after heart transplantation. While porcine models are widely used in translational research, the tolerable duration of cold storage within an auto transplantation setting remains undefined. This study aimed to characterize functional recovery after 1.5 h and 3 h of hypothermic ischemia and establish a reproducible porcine model for evaluating novel preservation strategies.
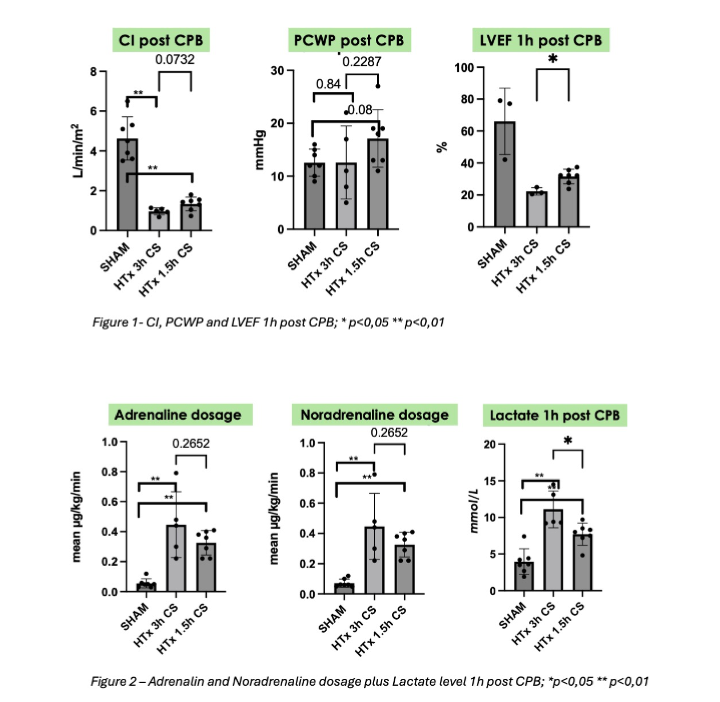
Methods
Nineteen pigs (mean 63 kg) were allocated to SHAM (n=7, cardiopulmonary bypass [CPB] only) or orthotopic auto transplantation in biatrial technique with either 1.5 h (n=7) or 3 h (n=5) of cold storage. Hemodynamic monitoring via pulmonary-artery-catheter included cardiac index (CI), pulmonary capillary wedge pressure (PCWP) and epicardial ultrasound-derived left ventricular ejection fraction (LVEF). Catecholamine requirements and lactate levels were assessed perioperatively. Data were analyzed with non-parametric tests (p<0.05).
Results
Cold ischemia impaired hemodynamics versus SHAM. CI significantly decreased after 1.5 h and 3 h storage versus SHAM (0.99 and 1.43 vs 4.5; p<0.05). LVEF 1 h post-reperfusion was lower after 3 h than 1.5 h (21% vs. 32%, p<0.05). PCWP showed no group difference (p=0.1952) (figure 1). Catecholamine doses were significantly higher after cold storage compared to SHAM (p<0.001) but similar between ischamic doses. Lactate levels were significantly elevated after 3 h versus 1.5 h (9.4 vs. 8.0 mmol/L, p<0.01) (figure 2). Two pigs in 3 h group failed to be weaned from CPB.
Conclusion
Both 1.5 h and 3 h of cold ischemia significantly impaired myocardial function versus SHAM. A duration of 1.5 h induced reproducible hemodynamic compromise and represents an optimal time frame to assess ischemic injury in this novel porcine auto transplantation model. This model reliably detects myocardial injury and provides a robust platform for testing new preservation strategies while reducing the number of animals required.
Molecular Profiling of Perfusate After Normothermic Ex Vivo Machine Perfusion to Identify Markers Associated with Primary Graft Dysfunction After Cardiac Transplantation
440 – Molecular Profiling of Perfusate After Normothermic Ex Vivo Machine Perfusion to Identify Markers Associated with Primary Graft Dysfunction After Cardiac Transplantation
Featured Poster Session 2 (Non-CME): Basic Science – Perfusion-Associated Gene Changes
Thursday, 23 April | 4:30 – 5:30 p.m. | Poster Theater 1

A. Alvarez1, G. Esmailian2, M. Mendiola Pla1, L. Chacon3, S. Casalinova4, K. Dewan2, R. Gross2, C. Wang2, J. Schroder2, C. Milano2, C. Hochman-Mendez5, D. Bowles6. 1Duke University, Durham, NC, 2Duke University Medical Center, Durham, NC, 3University of St Thomas, Houston, TX, 4Duke University Hospital, Durham, NC, 5The Texas Heart Institute, Houston, TX, 6Duke, Durham, NC.
Abstract
Purpose
Risk assessment for primary graft dysfunction (PGD) after cardiac transplantation currently depends on donor and recipient characteristics. Because the Organ Care System (OCS) perfusate reflects intrinsic donor organ biology before implantation, its molecular profiling may reveal markers associated with PGD. We evaluated microRNA (miR) and cytokine profiles in OCS perfusate to identify signatures linked to PGD incidence.
Methods
Perfusate samples (n=37) were collected after OCS heart perfusion from donation after brain death (DBD) and donation after circulatory death (DCD) donors. Samples were stratified by donor type and recipient outcome: DBD-PGD (n=7), DBD-non-PGD (n=10), DCD-PGD (n=10), and DCD-non-PGD (n=10). Cytokines were quantified using a bead-based multiplex immunoassay on the Luminex Intelliflex platform, and miRs were profiled from exosomal cargo isolated via membrane affinity-based purification followed by high-throughput small-RNA sequencing.
Results
Unbiased miR profiling and cytokine analysis revealed distinct molecular patterns between donor types. In DBD donors, PGD was associated with upregulation of miR-125a/b, consistent with inflammatory activation, along with increased interleukin-6, granulocyte colony-stimulating factor, and interleukin-10 levels. In DCD donors, PGD was characterized by downregulation of miR-122-5p concurrent with elevated lactate levels, suggesting a metabolic-stress signature.
Conclusion
Perfusate miR and cytokine profiling identified donor-intrinsic molecular signatures associated with PGD. DBD donors showed an inflammatory pattern, while DCD donors exhibited evidence of metabolic dysregulation. These findings highlight the potential of OCS perfusate profiling for pre-transplant PGD risk stratification and suggesting mechanistic differences between PGD arising from DBD and DCD hearts.
Post-Reperfusion Metabolic Implications of 10°C Static Preservation of Donor Lungs
441 – Post-Reperfusion Metabolic Implications of 10°C Static Preservation of Donor Lungs
Featured Poster Session 2 (Non-CME): Basic Science – Perfusion-Associated Gene Changes
Thursday, 23 April | 4:30 – 5:30 p.m. | Poster Theater 1

M. Martinez Santos1, A. Wang2, A. Ali2, M. Liu2, S. Keshavjee2, M. Cypel2. 1University of Toronto, Toronto, ON, Canada, 2Latner Thoracic Research Laboratories, Toronto General Hospital Research Institute, Toronto, ON, Canada.
Abstract
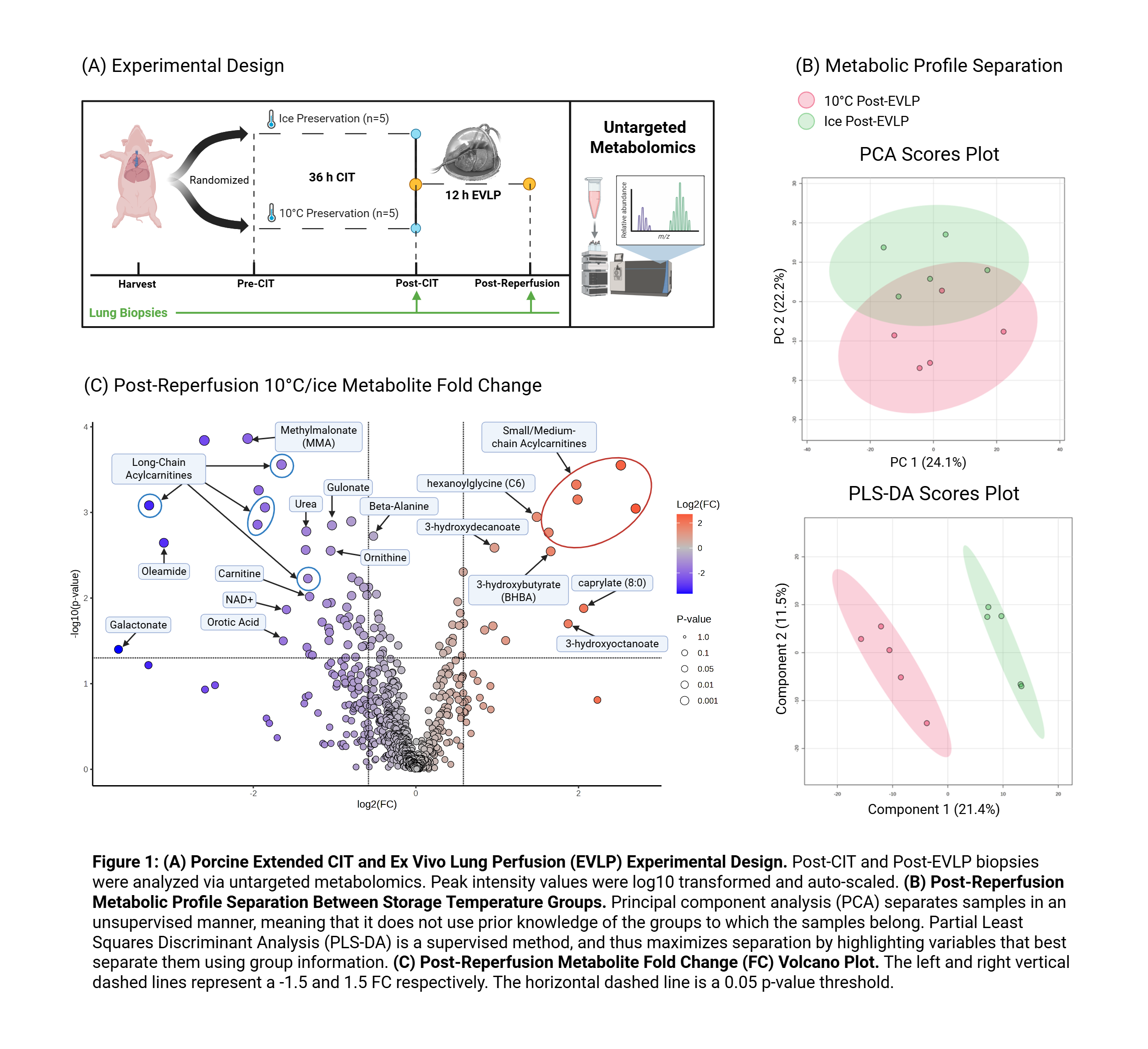
Purpose
The safe extension of donor lung cold ischemia time (CIT) at 10°C has been linked with mitochondrial resilience at this temperature, resulting in superior metabolic activity and graft viability during preservation. However, little is known about the impact of 10°C preservation on the metabolic profile of the reperfused grafts. We aimed to identify critical pathways that contribute to the functional benefits of 10°C lung storage, identify metabolites that may predict allograft resilience, and inform therapeutic targets to improve transplant outcomes.
Methods
Porcine donor lungs (n=5/group) were randomized to 36h of CIT on ice or 10°C and subsequently subjected to 12h of ex vivo lung perfusion (EVLP) (Figure 1A). Lung biopsies were obtained at the end of CIT and the end of EVLP for analysis via untargeted metabolomics using MetaboAnalyst6.0. Significantly different metabolites were identified and analyzed to elucidate the underlying molecular mechanisms.
Results
A clear tissue metabolic profile separation was observed between the temperature groups after reperfusion. (Figure 1B). 10°C preservation induced a greater reactivation of cytoprotective and energy conserving pathways (purine and pyrimidine recycling), decreased urea cycle activity, and greater capacity for aerobic respiration (as seen by levels of sugars and TCA cycle intermediates). (Figure 1C). Acylcarnitines accounted for the greatest difference, suggesting enhanced ꞵ-oxidation at both the mitochondria and peroxisomes after 10°C preservation.
Conclusion
Our study demonstrates that extended 10°C preservation primes lungs for superior metabolic recovery and allograft resilience during EVLP, likely contributing to reduced ischemia reperfusion injury and improved outcomes. The multi-center randomized-controlled clinical trial (NCT05898776) comparing 10°C to conventional ice preservation is set to finish enrollment this year. We will confirm our findings described herein in humans using biobanked tissue collected in this study.
Infectious Diseases
Poster Theater 2
The Impact of Intraoperative Hypothermia on Surgical Site Infections in Pediatric Heart Transplant
442 – The Impact of Intraoperative Hypothermia on Surgical Site Infections in Pediatric Heart Transplant
Featured Poster Session 2 (Non-CME): Infectious Diseases
Thursday, 23 April | 4:30 – 5:30 p.m. | Poster Theater 2

J. K. Kaniuk1, C. F. Bennett1, W. J. Muller2. 1Northwestern University Feinberg School of Medicine, Chicago, IL, 2Division of Infectious Diseases, Ann & Robert H. Lurie Children’s Hospital of Chicago, Chicago, IL.
Abstract
Purpose
Intraoperative hypothermia (IH) has been linked to increased risk of surgical site infections (SSI) in the adult population, but there is less known about IH in pediatric heart transplant (OHT). We hypothesized that IH would increase the risk of SSI in pediatric OHT.
Methods
We retrospectively reviewed pediatric OHT surgeries at our center between 2015 and 2025. Patient temperatures during OHT surgeries were stratified by groups based on lowest temperature achieved during the surgery using right nasopharyngeal temperatures from anesthesia reports. Temperature groups were not hypothermic (NH) (>95.0 °F), mild hypothermia (MiH) (89.7 °F - 95.0 °F), moderate hypothermia (MoH) (82.5 °F - 89.6 °F), and severe hypothermia (SH) (≤ 82.4 °F). The association of degree of hypothermia with post-operative infections within 30 days of surgery was assessed by multivariate logistic regression, controlling for age, race, sex, ethnicity, and total procedure time. Post-hoc pairwise comparison determined differences between hypothermia subgroups.
Results
There were 130 OHT procedures over the time period studied. Temperatures during OHT were NH in 5 patients (3.8%; 80.0% male; 80.0% white), MiH in 15 (12.0%; 73.3% male; 60% white), MoH in 59 (47.2%; 54.2% male; 42.4% white) and SH in 51 (40.8%; 49.0% male; 45.1% white). Infection rates were 60% in the NH cohort (3), 53.3% in the MiH cohort (8 infections), 55.9% in the MoH cohort (33 infections), and 82.4% in the SH cohort (42 infections). There was a statistically higher risk of infection for the SH cohort compared to MoH (OR = 0.79 [0.64-0.99], p =0.04) on multivariate logistic regression, but there was no difference in any other models (p > 0.05).
Conclusion
In our series of pediatric OHT procedures, IH did not significantly increase overall risk of SSI. However, there was an increased risk of SSI after SH compared with MoH. More work is needed to determine whether additional factors including nutritional status, hypothermia duration, or comorbidities have affect the association between IH depth and SSI.
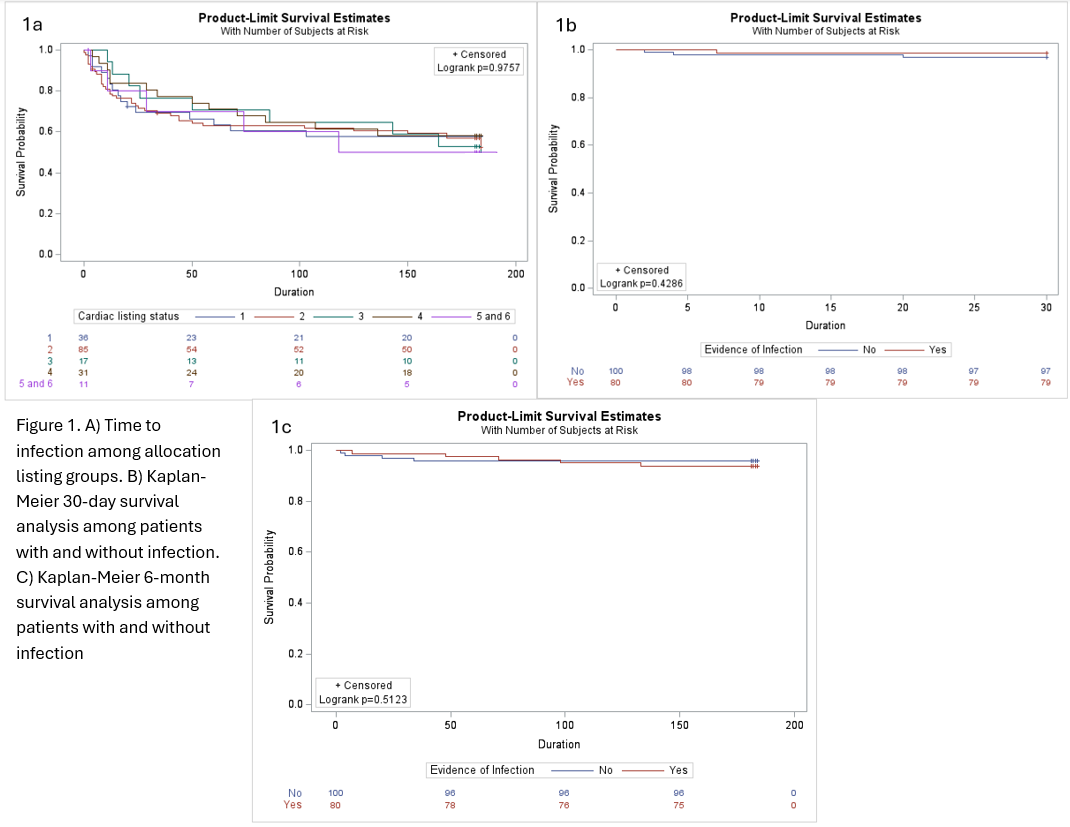
Higher Heart Transplant Allocation Priority Status is Not Associated with Increased Infection Risk in the Contemporary Era
443 – Higher Heart Transplant Allocation Priority Status is Not Associated with Increased Infection Risk in the Contemporary Era
Featured Poster Session 2 (Non-CME): Infectious Diseases
Thursday, 23 April | 4:30 – 5:30 p.m. | Poster Theater 2

A. De La Hoz Gomez, A. J. O'Neill, D. J. Regan, E. Woehl, G. C. Valentine, S. Gupta, A. S. Desai, M. Givertz, A. Itoh, R. Maurer, L. R. Baden, M. Mehra, A. E. Woolley. Brigham and Women's Hospital, Boston, MA.
Abstract
Purpose
In 2018, the US implemented a revised 6-tier heart transplant allocation system to prioritize patients with the highest urgency. This change has driven increasing use of mechanical circulatory support (MCS) as a bridge to transplantation. We assessed whether this epidemiological shift is associated with increased rates of post-transplant infection.
Methods
We conducted a retrospective study of adult heart transplant recipients (OHT) at our center from 2018 to 2025 . Patients were categorized by infection status within the 6-months following transplant. The relationship between allocation priority and time to first infection, and impact of infection on 30-day and 6-month survival were displayed using Kaplan-Meier plots and compared using log-rank tests.
Results
Among 180 OHT, 80 (40%) developed any infection within 6-months (median 23 days; IQR 9.5-59) (Table 1). Infections included bacteremia (30%), pulmonary (73%), sternal (24%), and abdominal (20%). 43 OHT (54%) had >1 infection type. The listing priority at the time of transplantation was similar between those with and without infection (p=0.88). A higher proportion of infections was noted in those receiving post-transplant temporary MCS (p=0.01) or with delayed chest closure (p=0.04). There were no significant differences in survival in those with and without infection (p=0.51) (Figure 1).
Conclusion
In this single center analysis, higher priority listing at the time of transplantation was not associated with increased risk of post-transplant infection. Survival did not differ in those with and without early postoperative infections.
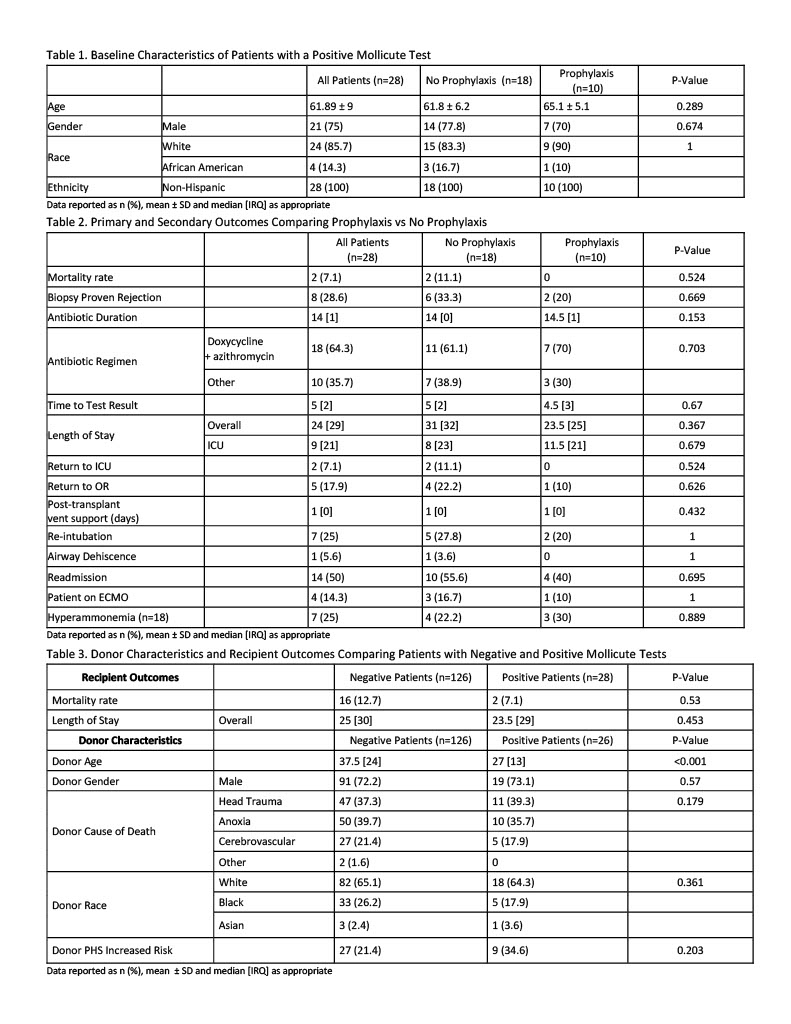
Universal Screening and Prophylaxis of Mycoplasma and Ureaplasma in Lung Transplant Patients
444 – Universal Screening and Prophylaxis of Mycoplasma and Ureaplasma in Lung Transplant Patients
Featured Poster Session 2 (Non-CME): Infectious Diseases
Thursday, 23 April | 4:30 – 5:30 p.m. | Poster Theater 2

S. Roussel, M. Czarnecki, J. Dann, J. Geyston, R. Anderson, M. Weder, H. Mannem. University of Virginia, Charlottesville, VA.
Abstract
Purpose
Donor-derived Mycoplasma and Ureaplasma pose a serious risk to lung transplant (LT) recipients. The University of Virginia Health implemented a novel screening strategy with polymerase chain reaction testing from intraoperative swabs of the donor bronchus, followed by universal prophylaxis (PPX). This study aims to evaluate the impact of universal screening (US) and PPX in LT patients.
Methods
This was a retrospective observational study. Patients with a lung transplant between Nov 2016 and Aug 2025, with Mycoplasma and/or Ureaplasma testing completed were included. US of donor lungs was initiated in Nov 2020. Positive patients were initiated on treatment at time of test result (for cause) or initiated on treatment at time of transplant (PPX). The primary outcome was mortality at 12 months in patients with a positive test, comparing for-cause versus PPX. Secondary outcomes compared a cohort of donors with a negative versus positive test for mortality, length of stay (LOS) and donor characteristics.
Results
253 patients were screened, and 28 patients had a positive test. 148 donors underwent US; there was a 14.9% prevalence. Median time to positive result was 5 days. Patients with positive tests were treated for a median of 14 days. Mortality and other posttransplant outcomes in patients with positive tests did not change if they received treatment or PPX (table 2). For patients with negative donor screening versus those with positive tests, mortality and LOS were similar. Donors with positive screening were significantly younger. In donors under the age of 35, prevalence of positivity increased to 24%.
Conclusion
Use of a novel OR back-table swab method offers a safe, non-invasive-to-recipient opportunity for US, allowing for safe PPX of Mycoplasma and Ureaplasma infections. There was no difference in mortality between patients with positive and negative tests, with no complications related to PPX. Younger donors were more likely to have a positive screening test leading to potential targeted PPX population.
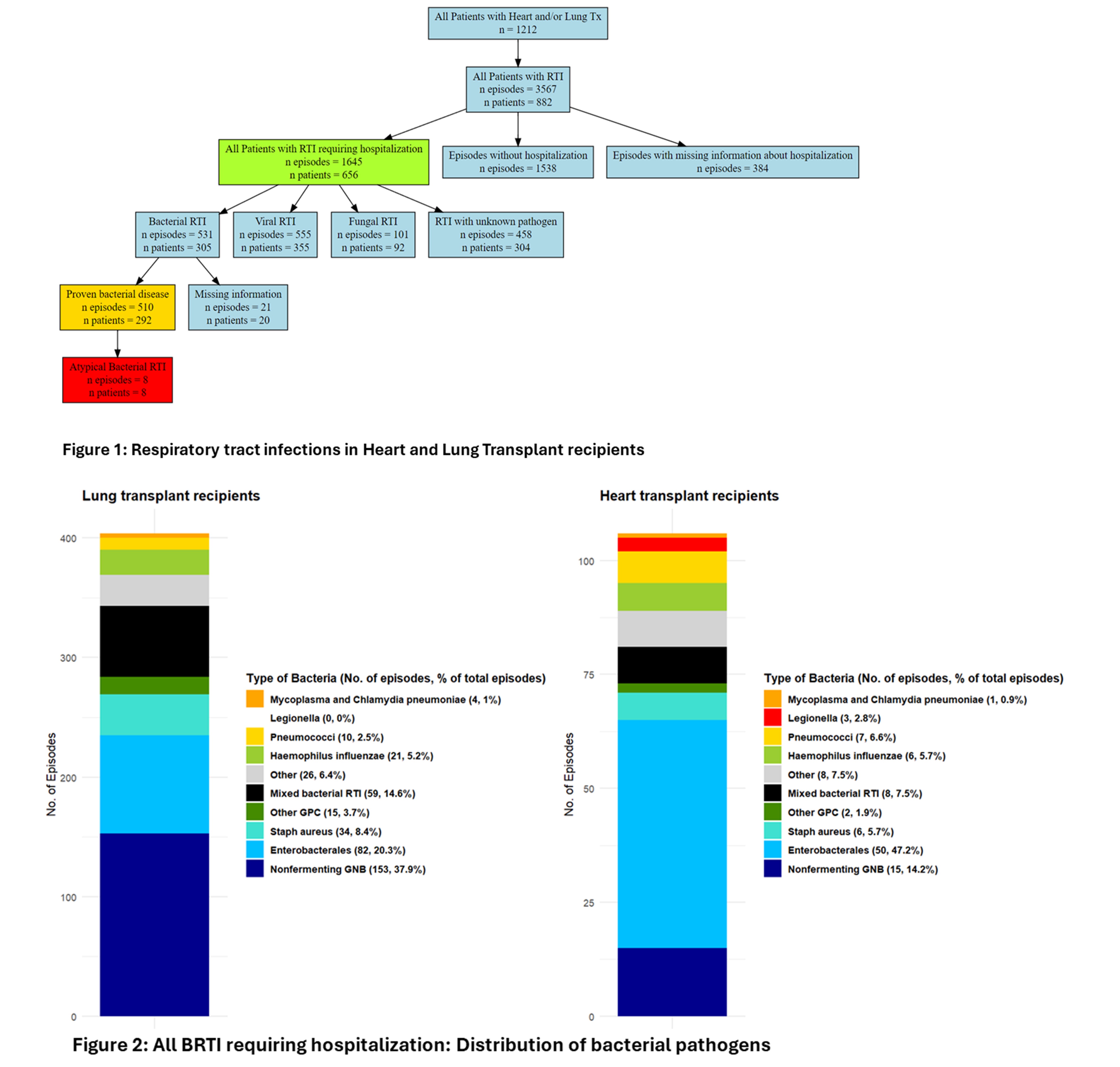
Atypical Bacterial Respiratory Pathogens in Thoracic Transplant Recipients - A Nation-Wide Study
445 – Atypical Bacterial Respiratory Pathogens in Thoracic Transplant Recipients - A Nation-Wide Study
Featured Poster Session 2 (Non-CME): Infectious Diseases
Thursday, 23 April | 4:30 – 5:30 p.m. | Poster Theater 2

J. Bisch-Karatas1, I. Abela2, B. Schnegg3, A. Koutsokera4, M. Schuurmans5, O. Manuel6, A. Flammer7, C. van Delden8, N. Mueller7, C. Hirzel1, L. N. Walti9. 1Inselspital, Bern, Bern, Switzerland, 2University Hospital Zürich, Zurich, Switzerland, 3Inselspital, University Hospital Bern, CH, Bern, Switzerland, 4Lausanne University Hospital, Lausanne, Switzerland, 5Univ of Zurich, Lung Transplant Medical Director, Zurich, Switzerland, 6Univ Hosp of Lausanne, Lausanne, Switzerland, 7University Hospital Zurich, Zurich, Switzerland, 8University Hospital of Geneva (HUG), Geneva, Switzerland, 9Inselspital Bern, Schweiz, Bern, Switzerland.
Abstract
Purpose
Bacterial respiratory tract infections (BRTI) are important contributors to morbidity in thoracic solid organ transplant (TSOT) recipients. In TSOT recipients, empiric treatment often covers “atypical” pathogens. However, their epidemiology, clinical presentation and outcomes are not well described.
Methods
We included all heart- and lung-recipients enrolled (2008-2023) in the Swiss Transplant Cohort Study and assessed epidemiology of clinically relevant bacterial respiratory infections with a focus on “atypical” pathogens (Legionella spp., Mycoplasma pneumoniae, Chlamydophila pneumoniae, Coxiella burnetii and Francisella tularensis) and investigated associated 90 day mortality.
Results
We included 1212 TSOT recipients with a follow-up of 6`909 person-years (py). Bacterial pathogens were identified in 510 episodes (31%) of clinically relevant respiratory infections (Figure 1). 24% (292/1212) suffered from at least one BRTI. The overall incidence of BRTI was 7382/100`000py. Lung- recipients had a higher incidence then heart (11298 vs. 3180/100`000py, p<0.001) and pathogen distribution differed (Figure 2): In both gram-negative bacteria dominated: Pseudomonas aeruginosa was the most prevalent pathogen in lung- (38%), Enterobacterales in heart-recipients (47%). BRTI were associated with an increased 90-days mortality (HR 5.92, 95% CI 4.08-8.60, p<0.001). Atypical pathogens were rarely found: We identified 3 cases of Legionella (all in heart-recipients, none on TMP-SMX prophylaxis) and 6 cases of Mycoplasma and Chlamydia pneumoniae. They occurred late after transplant (median 2.35 years) and were associated with frequent hospitalization (89%) but low ICU rates (11%), no patient died within 90 days.
Conclusion
BRTI were prevalent in TSOT recipients, and distribution depends on organ transplant with potential implications on empiric treatment. However, atypical bacterial pathogens were infrequently identified, despite frequent empiric coverage.
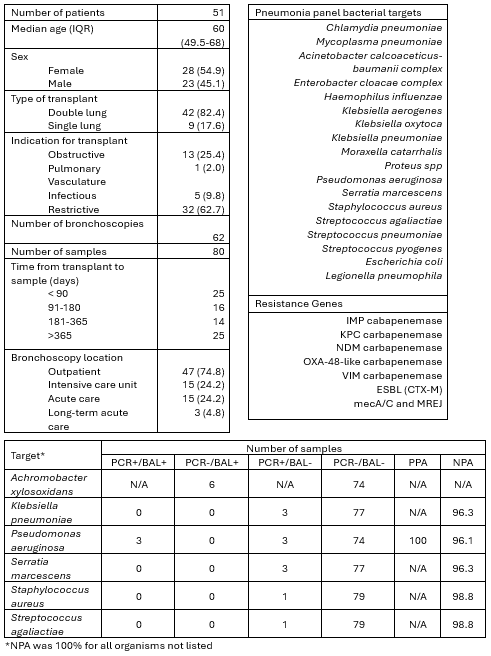
Comparison of Pneumonia Panel Polymerase Chain Reaction Pathogen Identification to Bronchoalveolar Lavage Culture in Lung Transplant Recipients
446 – Comparison of Pneumonia Panel Polymerase Chain Reaction Pathogen Identification to Bronchoalveolar Lavage Culture in Lung Transplant Recipients
Featured Poster Session 2 (Non-CME): Infectious Diseases
Thursday, 23 April | 4:30 – 5:30 p.m. | Poster Theater 2

A. T. Hutchins1, E. Rubido2. 1Pharmacy, Houston Methodist Hospital, Houston, TX, 2Houston Methodist Hospital.
Abstract
Purpose
Bronchoalveolar lavage (BAL) cultures are an important tool in the identification and antimicrobial sensitivity testing of pathogens causing lower respiratory tract infections. Molecular diagnostics such as the pneumonia panel polymerase chain reaction panel (PN panel) can supplement standard culture-based diagnostics to provide rapid identification of potential pathogens and resistance mechanism allowing for timely antibiotic tailoring and de-escalation. However, the use of the PNA panel is poorly described in lung transplant recipients (LTRs).
Methods
We reviewed PN panel and BAL bacterial culture results from LTRs who underwent bronchoscopy between September 8 and September 21, 2025. We compared results of PN panel to BAL bacterial culture to evaluate the positive percent agreement (PPA) and negative percent agreement (NPA) between tests.
Results
Fifty-one LTRs underwent 62 bronchoscopies resulting in 80 samples included for analysis. LTRs were primarily female (54.9%) double LTRs (82.4%) for restrictive lung disease (62.7%). Fourteen PNA panels and nine BALs were positive for bacterial pathogens. PPA was 100% for bacterial pathogens included in the PN panel. However, six cultures (7.5%) were positive for organisms not reported on the PN panel. NPA was >95% for all bacterial pathogens included in the PN panel.
Conclusion
There were high PPA and NPA between PN panel and BAL cultures in the sample population for pathogens detected by the PN panel. However, BAL cultures were positive for clinically relevant pathogens not detected by the PN panel that may have greater prevalence in LTRs. Further study is necessary to validate these findings and elucidate the role of PN panel in guiding antimicrobial utilization and stewardship in LTRs.
Featured Poster Session 03
Friday, 24 April | 4:30 – 5:30 p.m.
Basic Science - Biomarkers
Poster Theater 1
Identification of Population-Enriched Pathogenic TTN Variant Using Fine-Grained Genomic Ancestry
447 – Identification of Population-Enriched Pathogenic TTN Variant Using Fine-Grained Genomic Ancestry
Featured Poster Session 3 (Non-CME): Basic Science – Biomarkers
Friday, 24 April | 4:30
– 5:30 p.m. | Poster Theater 1

H. V. Gupta1, M. Isshiki1, I. Raghuram1, B. E. Ferrell2, M. Colón3, J. M. Greally1, A. Weissler-Snir4, S. Raj1. 1Department of Genetics, Albert Einstein College of Medicine, Bronx, NY, 2Cardiothoracic and Vascular Surgery, Montefiore Medical Center, Bronx, NY, 3Hondurans Against AIDS/Casa Yurumein, Bronx, NY, 4Department of Cardiology, Montefiore Medical Center, Bronx, NY
Abstract
Purpose
Even when moving beyond self-identified ancestry to genomic ancestry, enriched pathogenic variants may still be overlooked due to bucketing individuals by continental ancestry. Here, we utilize large-scale biobank resources (All of Us - AOU) to perform fine-grained ancestry analysis and discover potential population-enriched pathogenic variants (PVs).
Methods
Identity-by-descent (IBD) was used to cluster all participants in AOU v8 with array and whole-genome sequencing into fine-grained clusters. PVs from various databases were tested for population enrichment using Fisher’s test. Phenotypic enrichment in populations was determined using logistic regression, controlling demographic factors, and recruitment sites. In accordance with AOU policies to not report counts of <20 individuals, we mainly report frequencies.
Results
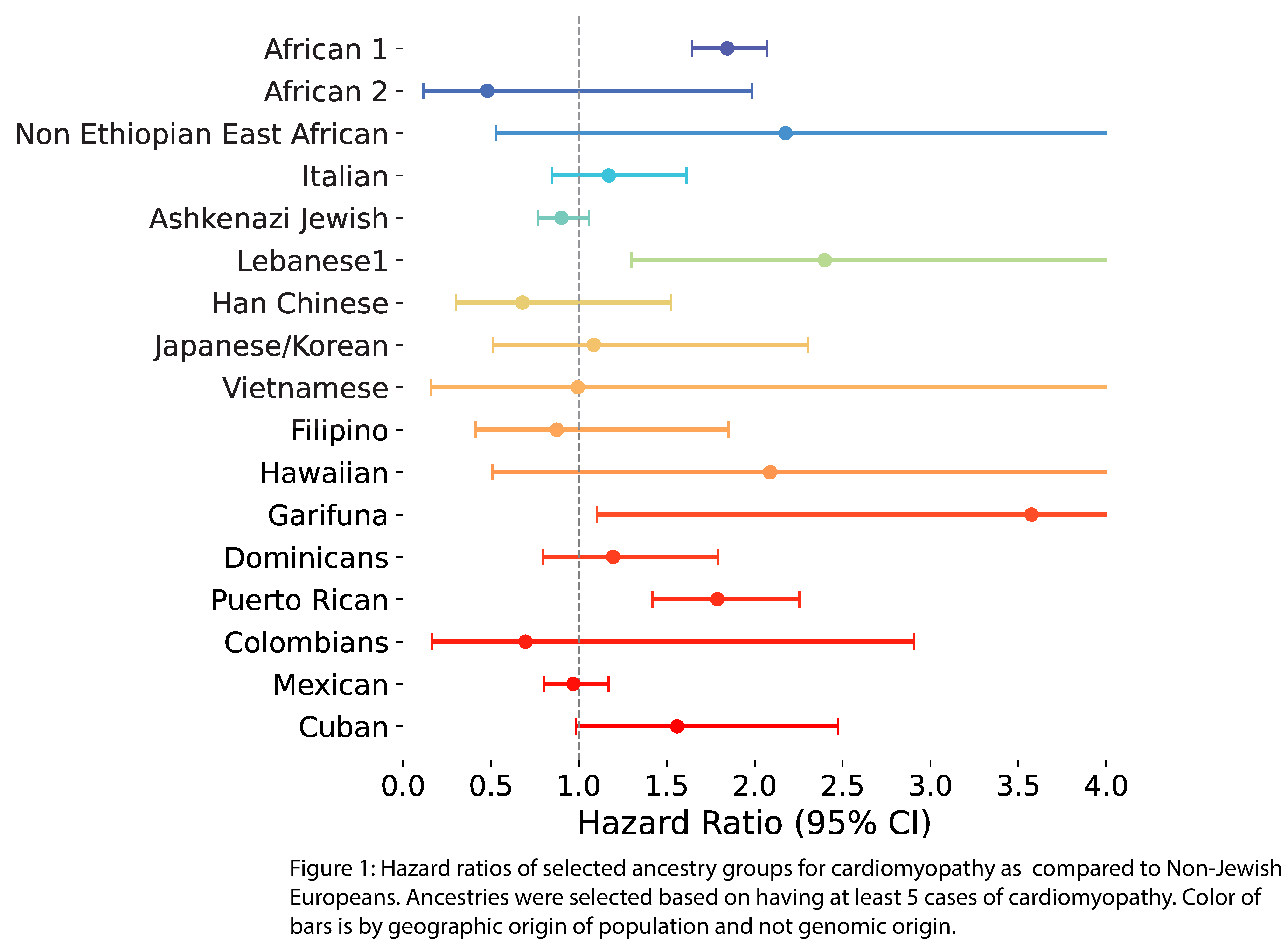
IBD clustering identified 58 clusters in 385k participants. A total of 340,883 PVs were tested for enrichment, using Bonferroni correction. The variant chr2:178542263:C:G, a PV in the gene TTN associated with dilated cardiomyopathy (DCM), was found more than once at a significantly enriched allele frequency of 0.6% in Garifuna individuals, while having a gnomAD frequency of <0.01% (adjusted p<0.001; Odds Ratio: 2451 Garifuna vs others). Using continental genomic ancestry, Garifuna with the PV were found to be in African ancestry, with non-significant enrichment of the variant (p=1.0, OR: 2.9). Garifuna had heightened odds of cardiomyopathy in All of Us (n = 3681 total), as compared to Non-Jewish Europeans (HR: 3.6 (1.1 - 11.6); p=0.03, Figure 1), with 100% penetrance of the variant for DCM.
Conclusion
We identify a population-specific PV in the Garifuna known to cause DCM and show matching clinical diagnoses. Future expansion of this work includes translational collaboration within our center and Garifuna representatives to screen Garifuna in our community, and expansion of efforts to discover relevant variation from fine-grained ancestry.
GRP78-Directed Autoantibodies (AABs) in BAL Are Associated with Future Chronic Lung Allograft Dysfunction (CLAD) and Define a CLAD-Specific Alveolar Type II (AT2) Phenotype Marked by the Unfolded Protein Response (UPR)
448 – GRP78-Directed Autoantibodies (AABs) in BAL Are Associated with Future Chronic Lung Allograft Dysfunction (CLAD) and Define a CLAD-Specific Alveolar Type II (AT2) Phenotype Marked by the Unfolded Protein Response (UPR)
Featured Poster Session 3 (Non-CME): Basic Science – Biomarkers
Friday, 24 April | 4:30
– 5:30 p.m. | Poster Theater 1

D. Vosoughi1, K. Bei1, A. Luca1, A. Chruscinski2, A. Duong1, S. Keshavjee3, T. Martinu1, S. Juvet1. 1Toronto Lung Transplant Program, University of Toronto, Toronto, ON, Canada, 2Toronto General Hospital, University of Toronto, Toronto, ON, Canada, 3Toronto Lung Transplant Program, UHN, Toronto, ON, Canada.
Abstract
Purpose
Serum AABs have been linked to CLAD; however, BAL AABs and their association with future CLAD has not been examined. It also remains unclear whether cell-surface antigens present on distinct cell subsets may be selectively targeted by an autoimmune response.
Methods
A multiplexed antigen microarray was used to profile 45 AABs in BAL at 3, 6, 12, 18, and 24-months (±14 days) post-LT from a case-control cohort of recipients who developed CLAD or remained CLAD-free by 5 years after transplant. Five-fold cross-validated backward selection identified the minimal antibody set most strongly associated with future CLAD. Explanted lungs from CLAD (n=12; 5 BOS, 7 RAS/mixed) or donor controls (CTRL, n = 6) were analyzed by flow cytometry (FC) and single-cell RNA sequencing (scRNA-seq). Five additional CLAD and CTRL samples were enriched for epithelial cells to validate phenotypes identified by scRNA-seq using FC.
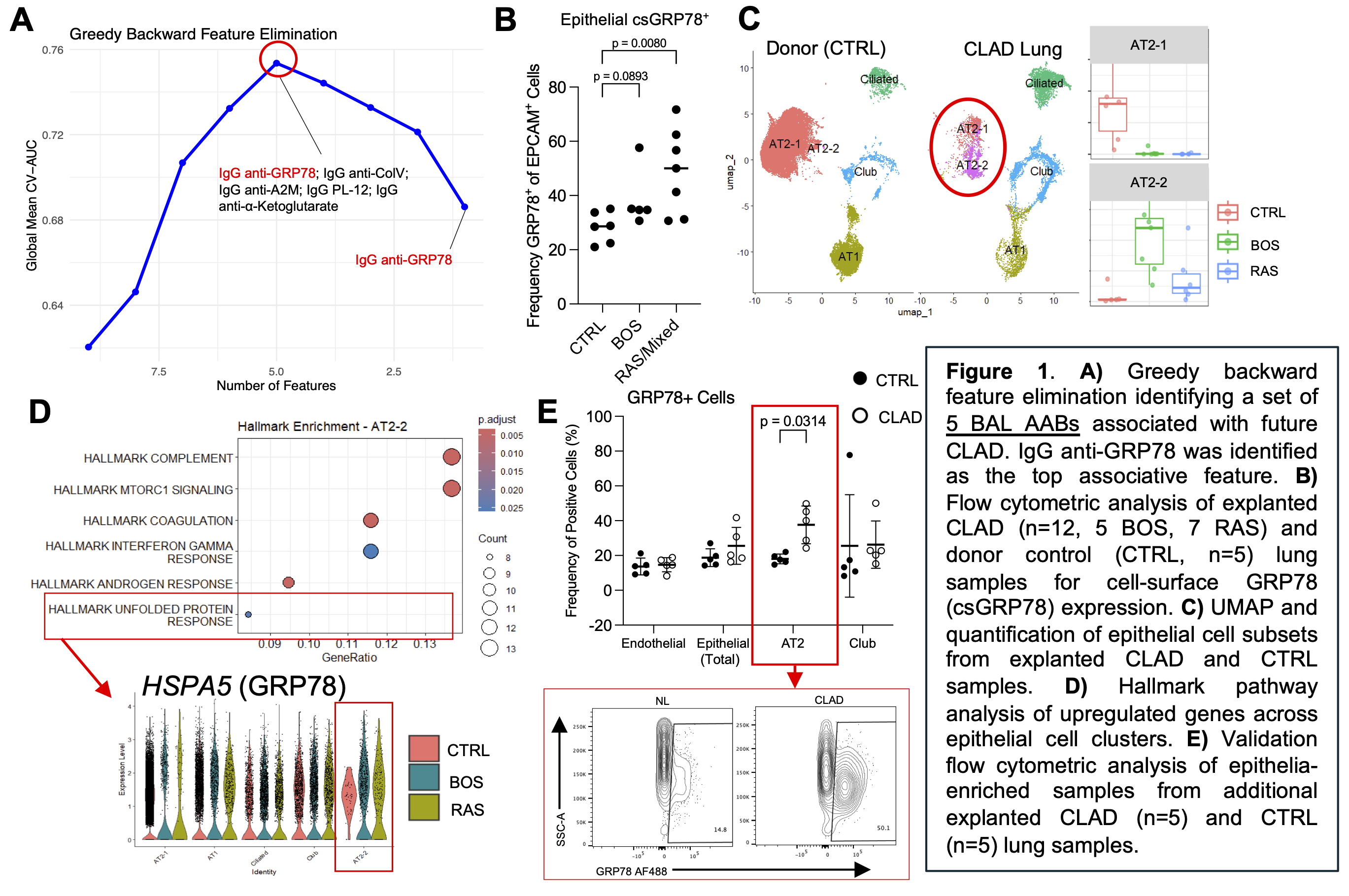
Results
Feature selection identified a combinatorial BAL signature comprised of 6 AABs associated with future CLAD (average AUROC = 0.753), with IgG anti-GRP78 emerging as the top associative feature (Fig.1A). The presence of its cognate antigen, GRP78, was significantly elevated on the cell-surface (csGRP78) of CLAD epithelia, especially in RAS/mixed (Fig.1B). On a transcriptional level, scRNA-seq identified a CLAD-specific AT2 subset (AT2-2, Fig.1C) with UPR pathway activation and HSPA5 upregulation (encoding GRP78, Fig.1D). FC of epithelia-enriched lung cells from additional samples validated the preferential csGRP78 expression on CLAD AT2 cells (Fig.1E).
Conclusion
BAL IgG anti-GRP78 was associated with future CLAD, with its cognate antigen marking an AT2 cell population characterized by UPR activation and csGRP78. This suggests that AT2-cell stress mediates csGRP78 expression, and that AT2-cell loss may both drive and result from local autoimmunity in CLAD.
Tissue Fibroblast Activation Protein as an Early Diagnostic Marker and Predictor of Chronic Lung Allograft Dysfunction
449 – Tissue Fibroblast Activation Protein as an Early Diagnostic Marker and Predictor of Chronic Lung Allograft Dysfunction
Featured Poster Session 3 (Non-CME): Basic Science – Biomarkers
Friday, 24 April | 4:30
– 5:30 p.m. | Poster Theater 1

J. Kaniuk1, Y. Miyashita1, T. Kaiho1, H. Nagata2, T. Nagano1, T. Hiroshima2, Y. Sugiura2, M. E. Kelly1, Y. Yan1, H. Sun1, T. Kimura2, A. Bharat1, T. Kanou2, Y. Shintani2, C. Kurihara1. 1Northwestern University, Chicago, IL, 2Osaka University, Osaka, Japan.
Abstract
Purpose
Chronic lung allograft dysfunction (CLAD) is the major cause of late graft failure after lung transplantation. Fibroblast activation protein (FAP), selectively expressed on activated fibroblasts, may reflect ongoing fibrotic remodeling and serve as an early diagnostic biomarker.
Methods
We analyzed FAP expression across murine lung transplant models, human CLAD tissue, and a clinical cohort. Single-cell RNA sequencing identified FAP expression patterns in two murine models (C57BL/6→C57BL/10 and BALB/c→C57BL/6) and in human lung tissue (CLAD n = 5; control n = 5). Immunohistochemistry with AI-based image quantification was performed on transbronchial biopsies from 240 recipients (62 CLAD, 178 non-CLAD). ROC analysis, Kaplan-Meier curves, and Cox regression evaluated diagnostic and prognostic performance.
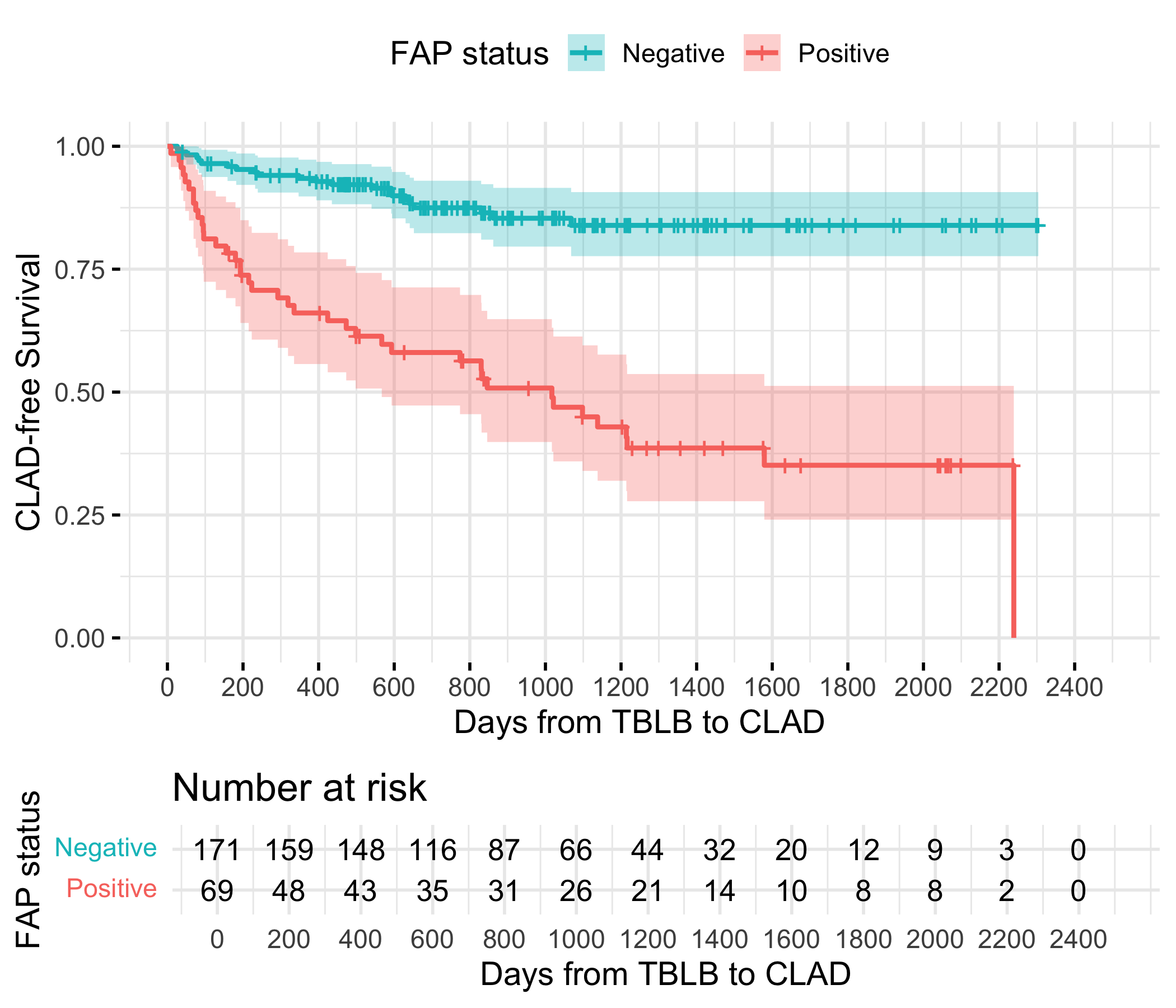
Results
FAP expression was confined to pathological fibroblast subsets and significantly increased during CLAD-associated remodeling in both murine and human lungs. Pathological and adventitial fibroblasts showed the highest FAP expression. In clinical samples, pre-CLAD biopsies had higher FAP-positive area than non-CLAD controls (13.0% vs. 5.1%, p < 0.0001). ROC analysis identified a 10.8% FAP threshold discriminating CLAD (AUC = 0.78; sensitivity 65%, specificity 84%). FAP-positive patients had shorter CLAD-free survival (p < 0.0001) and overall survival (p = 0.03). On multivariable Cox analysis, FAP positivity independently predicted CLAD (HR 5.43, 95% CI 3.22-9.16, p < 0.001).
Conclusion
FAP marks pathogenic fibroblast activity and predicts CLAD before clinical onset. Tissue FAP quantification can serve as a powerful tool for early diagnosis and risk stratification. Combining FAP staining with AI-based image analysis or molecular imaging may improve post-transplant surveillance and guide timely intervention.
Spatial Transcriptomics Reveals Coordinated Endothelial and Epithelial Activator Protein 1 Activation in Chronic Lung Allograft Dysfunction
450 – Spatial Transcriptomics Reveals Coordinated Endothelial and Epithelial Activator Protein 1 Activation in Chronic Lung Allograft Dysfunction
Featured Poster Session 3 (Non-CME): Basic Science – Biomarkers
Friday, 24 April | 4:30
– 5:30 p.m. | Poster Theater 1

Y. Goda1, M. Banday1, A. Potter2, R. Fernandez1, E. Suarez1, G. Loor1, D. Hayes2, N. Sharma1. 1Baylor College of Medicine, Houston, TX, 2Cincinnati Children's Hospital Medical Center, Cincinnati, OH.
Abstract
Purpose
CLAD remains a major issue in lung transplantation. This study aimed to characterize compartment-specific transcriptional profiles and their interactive pathways within epithelial, endothelial, and lymphocytes of CLAD, non-CLAD, and normal lungs.
Methods
Spatial transcriptomic profiling was performed using the GeoMx® Digital Spatial Profiler with Human Whole Transcriptome Atlas probes (>18,000 genes). Lung explants were obtained from CLAD (n=5), non-CLAD (n=3), and normal donor (n=3) samples. Regions of interest were classified as epithelium, endothelium, CD45⁺ lymphocyte aggregates, or bulk tissue. Differentially expressed genes (DEGs) were identified (FDR<0.05), followed by KEGG pathway enrichment and network analyses.
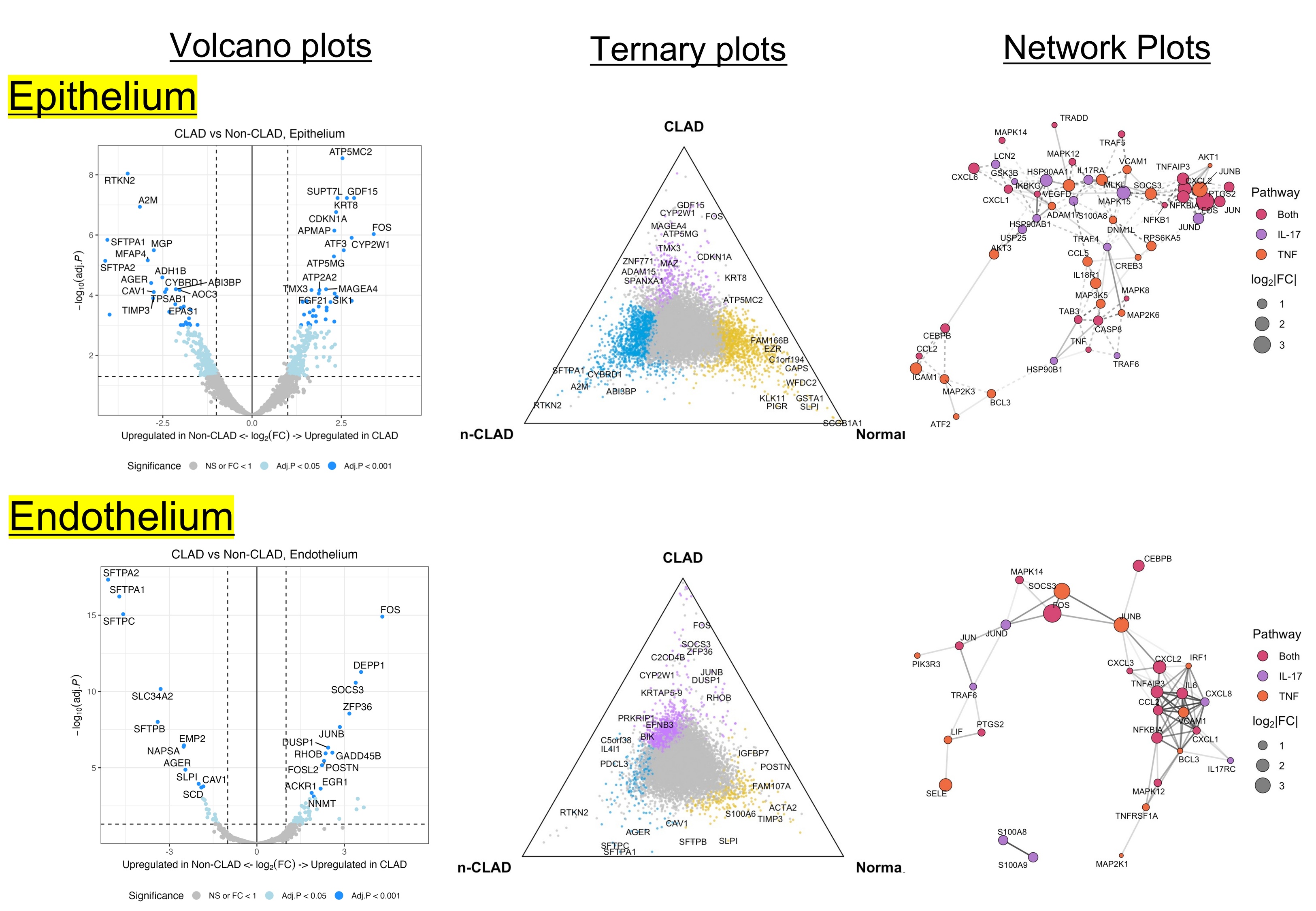
Results
CLAD lungs showed markedly higher DEGs in the epithelium than in the endothelium, with significant upregulation of AP-1 target genes JUNB and FOS. Conversely, A2M, an anti-fibrotic protease inhibitor, and surfactant proteins (SFTPA1/2) were downregulated, indicating epithelial dedifferentiation. Both epithelial and endothelial cells in CLAD exhibited distinct transcriptomic shifts characterized by increased inflammatory genes (CXCL1/2, IL6, SOCS3) and suppression of homeostatic pathways regulating barrier integrity (CLDN18, JAM3), antioxidant defense (GPX3), and protease balance (↑MMP7, ↓A2M). KEGG analysis revealed enrichment of IL-17, TNF, NF-κB, and ferroptosis pathways in CLAD, while non-CLAD epithelia showed protective signaling (PI3K-AKT, Rap1, MAPK, PPAR). Network mapping positioned JUNB and FOS as central hubs linking inflammation and fibrosis via NFKB1, MAPK1, and SOCS3.
Conclusion
Spatial transcriptomics revealed coordinated AP-1 activation in epithelial and endothelial compartments of CLAD lungs, driving pro-inflammatory and pro-fibrotic programs. Both compartments actively contribute to CLAD pathogenesis, highlighting vascular involvement in allograft failure.
Molecular Signatures Underlying Progression from Stage C to Stage D in Non-Ischemic Cardiogenic Shock
451 – Molecular Signatures Underlying Progression from Stage C to Stage D in Non-Ischemic Cardiogenic Shock
Featured Poster Session 3 (Non-CME): Basic Science – Biomarkers
Friday, 24 April | 4:30
– 5:30 p.m. | Poster Theater 1

B. Trethowan1, R. Loyaga-Rendon2, B. Chen3, R. Shankar4, A. Goodyke5, S. Koirala6. 1Cardiothoracic Critical Care, Corewell Health, Grand Rapids, MI, 2Advanced Heart Failure, Director of Research, Heart and Vascular Institute, Corewell Health, Grand Rapids, MI, 3Research, Michigan State University, Grand Rapids, MI, 4Michigan State University, Grand Rapids, MI, 5Translational Research Core, Corewell Health, Grand Rapids, MI, 6Department of Pediatric and Human Development, College of Human Medicine, Michigan State University, Grand Rapids, MI.
Abstract
Purpose
Cardiogenic shock (CS) is a progressive condition in which impaired cardiac function leads to multiorgan dysfunction and high mortality. The molecular mechanisms underlying CS progression remain poorly defined. Transcriptomic profiling of peripheral blood can provide insight into disease mechanisms and potential therapeutic targets. We aimed to characterize transcriptional differences between Stage C and Stage D non-ischemic CS patients.
Methods
Peripheral blood samples were collected from Stage C (n = 31) and Stage D (n = 9) non-ischemic CS patients within 24 hours of diagnosis. Bulk RNA sequencing was performed to generate gene expression profiles. Differentially expressed genes (DEGs) were identified by comparing Stage D with Stage C samples. Gene set enrichment analysis was conducted to identify biological pathways. Potential therapeutic compounds were inferred using the Reversal of Gene Expression Score (RGES) framework. Immune cell composition across stages was estimated using the CIBERSORTx. Survivors/non-survivors were compared.
Results
Differential expression analysis identified 87 DEGs (67 upregulated and 20 downregulated) in Stage D vs Stage C. Upregulated genes included MTND2P28, OSBPL1A, PEX11G, SEC14L5, and KCNMA1, while downregulated genes included TRGV10, ACOT11, and NDFIP2. Pathway analysis revealed activation of coagulation, platelet activation, and hemostasis pathways, alongside suppression of adaptive and T cell mediated immune responses, ribosome biogenesis, and rRNA processing. RGES analysis identified several candidate compounds, such as the JAK inhibitor upadacitinib and NSAIDs including ibuprofen and fenofibrate as potential therapies for Stage D. Cellular deconvolution revealed reductions in CD8 T cells, NK cells, CD4 memory cells, mast cells, and monocytes. Additionally, naïve B cells and M0 macrophages were reduced in deceased patients compared to survivors in Stage C group.
Conclusion
Transcriptomic profiling revealed distinct molecular signatures distinguishing Stage D from Stage C non-ischemic CS, characterized by activation of pro-thrombotic and oxidative stress pathways and suppression of adaptive immunity. Reduced immune cell abundance further suggests significant immune dysregulation in Stage D patients, providing insights into potential biomarkers and therapeutic targets for advanced CS.
Pathology
Poster Theater 2
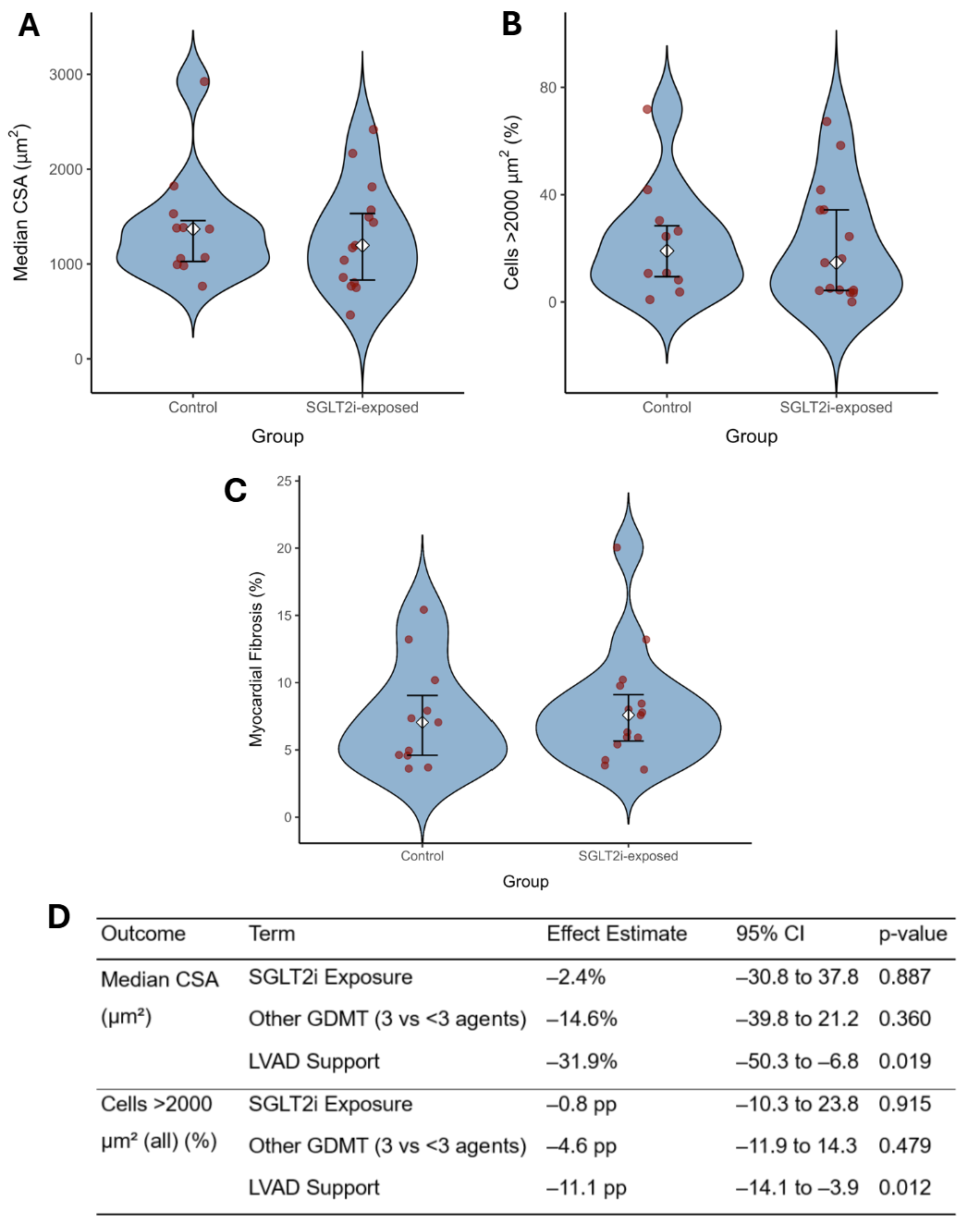
The Histological Impact of Sodium-Glucose Cotransporter-2 (SGLT2) Inhibitors in Patients with Non-Ischaemic Cardiomyopathy
452 – The Histological Impact of Sodium-Glucose Cotransporter-2 (SGLT2) Inhibitors in Patients with Non-Ischaemic Cardiomyopathy
Featured Poster Session 3 (Non-CME): Basic Science – Biomarkers
Friday, 24 April | 4:30
– 5:30 p.m. | Poster Theater 2

S. Bhatia1, H. Eckford2, X. Zhang3, V. Janbhandu4, L. Raven2, J. Greenfield2, C. Hayward2, V. Sivasubramaniam2, K. Muthiah2. 1University of New South Wales, Sydney, Australia, 2St Vincent's Hospital, Sydney, Australia, 3Stats Central, Mark Wainwright Analytical Centre, University of New South Wales, Sydney, Australia, 4Victor Chang Cardiac Research Institute, Sydney, Australia.
Abstract
Purpose
Sodium-glucose cotransporter-2 inhibitors (SGLT2i) improve outcomes in heart failure (HF), yet their cardioprotective mechanisms remain unclear. Myocardial fibrosis and hypertrophy are proposed targets but have not been histologically examined in humans.
Methods
Formalin-fixed left ventricular (LV) tissue from patients undergoing transplantation for non-ischaemic cardiomyopathy (NICM) was stratified by prior SGLT2i exposure. Fibrosis was quantified on polarised picrosirius red imaging and cardiomyocyte cross-sectional area (CSA) on haematoxylin and eosin staining, with the proportion of hypertrophic cells > 2000 µm² calculated. Group comparisons used Mann-Whitney U tests, and associations with SGLT2i exposure were evaluated using log-transformed multiple linear regression.
Results
Twenty-six LV samples were analysed (15 SGLT2i-exposed, 11 unexposed). Digital fibrosis quantification correlated strongly with pathologist estimates (Spearman ρ = 0.86) and inter-observer CSA agreement was high (ICC = 0.87). Median CSA did not differ between SGLT2i-exposed and control groups (1197 µm² vs 1369 µm²; p = 0.721; Figure A), nor did the proportion of hypertrophic cells (14.6% vs 19.0%; p = 0.646; Figure B) or fibrosis (7.6% vs 7.1%; p = 0.678; Figure C). After adjustment for LV assist device (LVAD) support and other guideline-directed medical therapy (GDMT), SGLT2i exposure remained unassociated with histological indices, whereas LVAD support predicted smaller cardiomyocytes (-31.9%; p = 0.019) and fewer hypertrophic cells (-11.1 pp; p = 0.012) (Figure D).
Conclusion
In advanced NICM, prior SGLT2i exposure was not associated with measurable reductions in LV fibrosis or hypertrophy. These findings suggest that structural remodelling may be less responsive to SGLT2i in end-stage HF, warranting further investigation in earlier disease stages and with longer treatment duration to determine whether structural benefits may emerge over time.
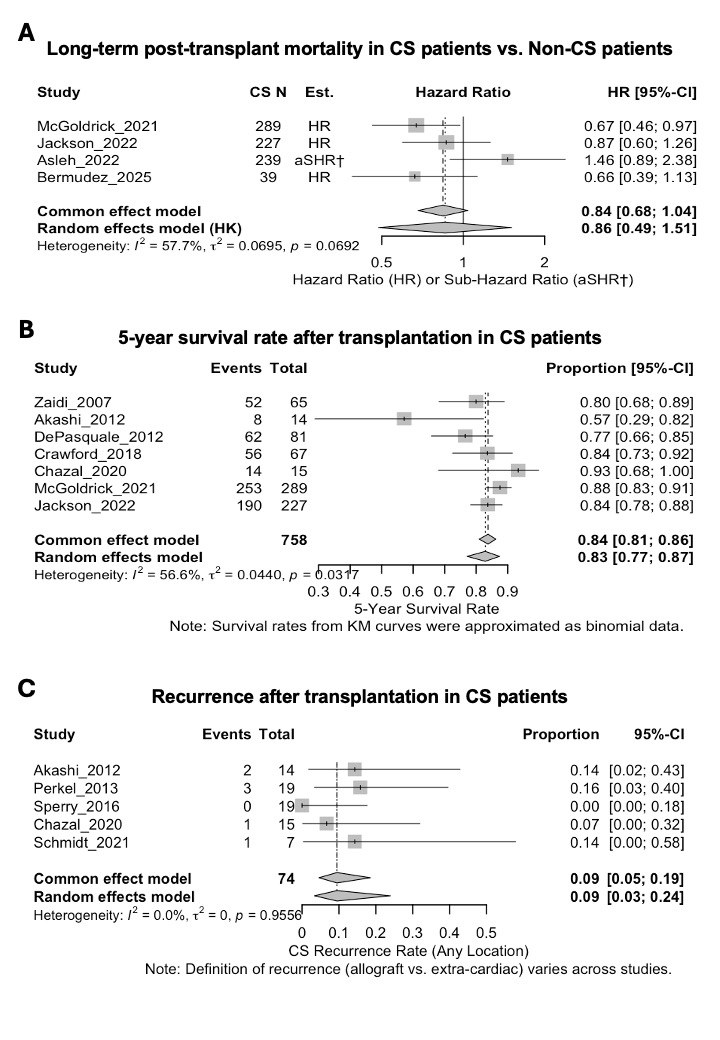
Long-Term Prognosis and Recurrence After Heart Transplantation in Cardiac Sarcoidosis: A Systematic Review and Meta-Analysis
453 – Long-Term Prognosis and Recurrence After Heart Transplantation in Cardiac Sarcoidosis: A Systematic Review and Meta-Analysis
Featured Poster Session 3 (Non-CME): Basic Science – Biomarkers
Friday, 24 April | 4:30
– 5:30 p.m. | Poster Theater 2

T. S. Kato1, D. Zhehestovska2, H. Sugiyama3, T. Yanai1, K. Shibao1, D. Akagaki1, K. Okabe1, Y. Fukumoto1. 1Dpt of Internal Medicine, Division of CV Medicine, Kurume University School of Medicine, Kurume, Fukuoka, Japan, 2Dpt of Internal Medicine, Division of CV Medicine, Kurume University School of Medicine, Kurume, Fukuoka, Japan; Department of Therapeutics and Family Medicine, Faculty of Postgraduate Education, Ivan Horbachevsky Ternopil National Medical University, Ternopil, Ukraine, 3Medical Student, Osaka University, Suita, Osaka, Japan.
Abstract
Purpose
Cardiac sarcoidosis (CS) is a progressive inflammatory cardiomyopathy that can cause life-threatening arrhythmias and advanced heart failure. Heart transplantation (HTx) is a treatment option for end-stage CS; however, its prognostic impact remains uncertain due to limited evidence and small cohort sizes.
Methods
A systematic literature search was conducted using PubMed, Scopus, and the Cochrane Library through October 2025 for studies reporting post-HTx outcomes in CS. Two reviewers independently performed screening, data extraction, and bias assessment (ROBINS-I). The primary outcome was all-cause mortality, and secondary outcomes included 5-year survival and post-HTx sarcoidosis recurrence. Random-effects meta-analysis was applied, and the certainty of evidence was graded using the GRADE approach.
Results
Thirteen observational studies met inclusion criteria. Long-term mortality after HTx in CS was comparable to that in non-CS cardiomyopathies (pooled HR 0.86, 95% CI 0.49-1.51, I²=57.7%: Figure A). The pooled 5-year survival rate was 83% (95% CI 77-87%, I²=56.6%: Figure B), and post-HTx recurrence incidence was 9% (95% CI 3-24%: Figure C). Evidence certainty for the primary outcome was very low due to the observational design and study heterogeneity.
Conclusion
Current evidence suggests that post-transplant outcomes in patients with cardiac sarcoidosis are comparable to those in other cardiomyopathies, with a relatively low recurrence rate after transplantation. These findings support the feasibility of HTx as an effective treatment for end-stage CS, while highlighting the need for high-quality, multicenter prospective studies to establish stronger evidence.
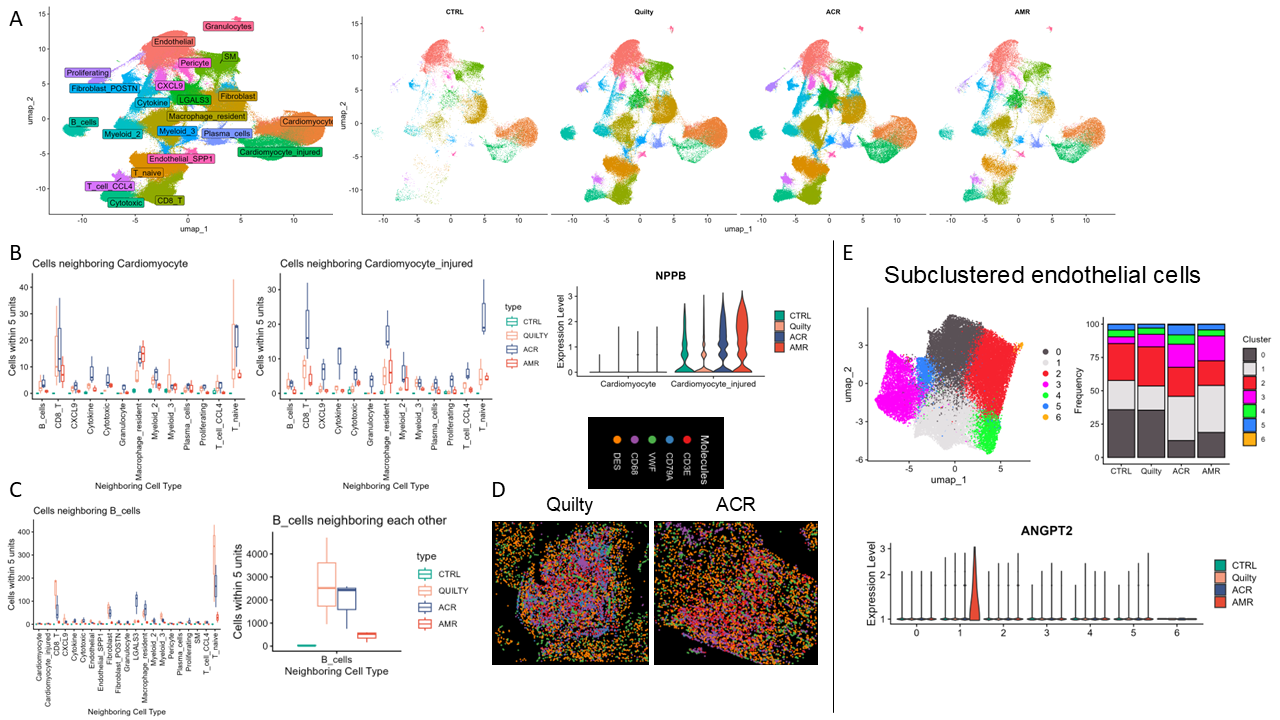
Spatial Transcriptomic Landscape of Acute Cellular Rejection, Antibody Mediated Rejection and Quilty Lesion of the Cardiac Allograft
454 – Spatial Transcriptomic Landscape of Acute Cellular Rejection, Antibody Mediated Rejection and Quilty Lesion of the Cardiac Allograft
Featured Poster Session 3 (Non-CME): Basic Science – Biomarkers
Friday, 24 April | 4:30
– 5:30 p.m. | Poster Theater 2

M. L. Axelrod1, K. Lavine2, C. Lin3. 1Pathology and Immunology, Washington University in St. Louis, St. Louis, MO, 2Washington University, St. Louis, MO, 3Pathology and Immunology, Washington University in St. Louis, St Louis, MO.
Abstract
Purpose
Acute cellular rejection (ACR) and antibody mediated rejection (AMR) are major immunological adverse events for cardiac transplant recipients, whereas Quilty lesion is considered a benign/reactive inflammatory infiltration. ACR, AMR and Quilty all contain admixed inflammatory cells and may be associated with cardiomyocyte damage. We aim to characterize the immunoprofile and cardiomyocyte characteristics of these three morphologically overlapping yet clinically distinct entities with single cell resolution.
Methods
With IRB approval, retrospective endomyocardial biopsies with ACR (n=3; grade 2R/3A by ISHLT 2004/1990 criteria), AMR (n=3; grade pAMR2), Quilty (n=3), and non-rejection (control, n=3) were identified. The samples were profiled using the 10X Genomics Xenium platform with the human multi-tissue panel with 100 custom add-on genes (total 477 genes). Data were analyzed in R using Seurat. Spatial neighboring relationships were characterized by calculating the distances between cells in populations of interest.
Results
Louvain clustering identified 22 clusters which were annotated based on differential gene expression (Figure 1A). The Cardiomyocyte_injured cluster was more prevalent in ACR and AMR than Quilty or control. Cells in the Cardiomyocyte_injured cluster also neighbored immune cells, especially CD8 T cells, in ACR (Figure 1B). Interestingly, there were similar proportions of immune clusters across ACR, AMR and Quilty; however, with differential gene expression and spatial localization. B cells in Quilty were present in aggregates with other B cells and naïve T cells, while they were more dispersed in ACR (Figure 1 C, D). Deeper characterization of endothelial cells identified a cluster, which uniquely expressed ANGPT2 in AMR (Figure 1E).
Conclusion
Spatial transcriptomics provides new insights into the pathogenesis of lesions of the cardiac allograft, and has the potential to identify novel disease features.
Extended Ex Vivo Lung Perfusion Does Not Increase Apoptotic Activity in a Swine Transplantation Model
455 – Extended Ex Vivo Lung Perfusion Does Not Increase Apoptotic Activity in a Swine Transplantation Model
Featured Poster Session 3 (Non-CME): Basic Science – Biomarkers
Friday, 24 April | 4:30
– 5:30 p.m. | Poster Theater 2

G. Cannone1, G. Maggioni2, A. Rebusso1, V. Verzeletti1, A. Bonis1, L. Lione1, G. Comacchio1, F. Calabrese2, A. Dell'Amore1, M. Schiavon1. 1Thoracic Surgery and Lung Transplant Unit, University of Padova, Padova, Italy, 2Anatomic Pathology Unit, University of Padova, Padova, Italy.
Abstract
Purpose
Organ Care System (OCS) is effective in improving lung condition with average perfusion times around 10 hours. Extending perfusion could allow longer reconditioning with possibility of targeted treatments. However, concerns remain about the potential for increased cellular injury over prolonged durations.
Methods
Porcine lungs preserved for 6h and 24h (10 each group) were included in the study. At the end of the perfusion, the right lung was analyzed to evaluate post perfusion effects and the left lung was transplanted in a recipient pig. Lungs were deemed transplantable based on perfusion parameters and final PaO2/FiO2 (P/F) > 300 mmHg. Post reperfusion (PR) selective (left pulmonary veins) blood gases at 1-2-3 hours were performed in addition to a left upper and lower lung biopsy at 1 hour (early RP sample). At 3 hours the animal was sacrificed, and the left lung sampled (late PR sample). Ischemia-reperfusion injury was analyzed on wet-to-dry ratio (W/D), histology (Fig 1A), TUNEL assay and immunohistochemistry (IHC).
Results
All preserved lungs were transplanted. OCS perfusion parameters were stable and comparable in both groups with similar median W/D (p=0.14). After transplantation, higher median P/F were observed in the 24h group. In none of the samples was found a significant difference between the two groups for the 8 histological parameters analyzed and TUNEL assay showed no differences in overall apoptotic levels (p=0.776) for every sample (Figure 1B). IHC revealed equivalent Caspase-8 levels (p=0.65, Fig 1C) and significantly higher Glutathione-Peroxidase 4 (GPX4) expression (Fig 1D) in the 6h group (p=0.0026).
Conclusion
Prolonged normothermic lung perfusion (24h) with OCS, showed comparable outcomes to shorter perfusion time (6h) and is not associated with increased apoptotic activity, supporting the feasibility of extended perfusion. The differential expression of GPX4 highlights potential time-dependent changes in antioxidant responses that warrant further investigation.
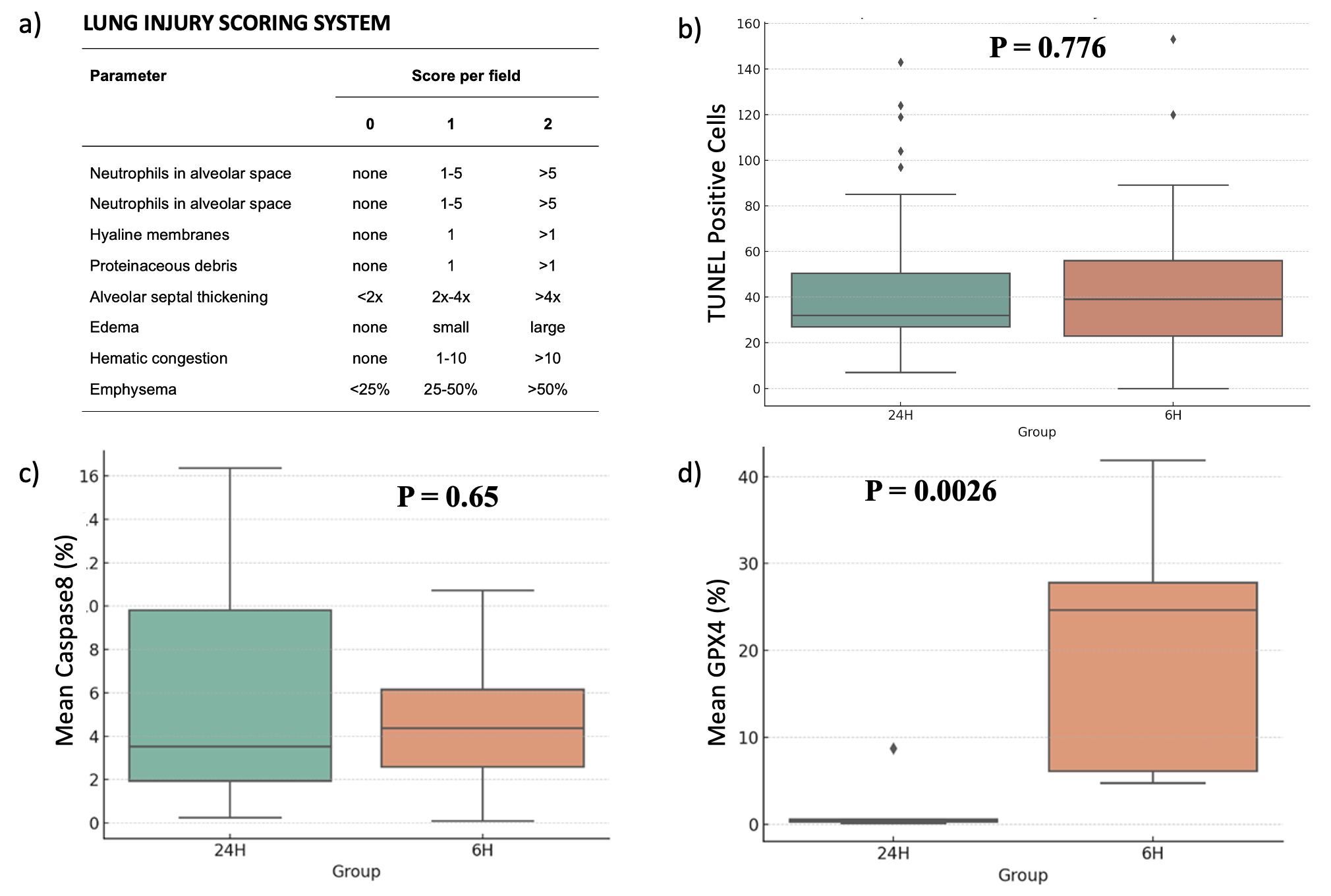
BOS and RAS Subtypes Differ in Explant Tissue Immune Subpopulations by Spatial Proteomics
456 – BOS and RAS Subtypes Differ in Explant Tissue Immune Subpopulations by Spatial Proteomics
Featured Poster Session 3 (Non-CME): Basic Science – Biomarkers
Friday, 24 April | 4:30
– 5:30 p.m. | Poster Theater 2

V. Vaira1, I. Righi2, L. Rosso3, N. Mansour3, M. R. De Filippo4, V. Andrea3, S. Brich5, D. Lorenzini3, G. Pruneri3, A. M. Storaci3, S. Franzi4, L. Morlacchi6, F. Blasi3, D. Trabattoni3, J. Greenland7, M. Nosotti3, S. Bosari3, M. Clerici8. 1University of Milan, Milano, Italy, 2Fondazione IRCCS Cà Granda Ospedale Maggiore Policlinico di Milano, Milan, Italy, 3University of Milan, Milan, Italy, 4Fondazione IRCCS Ca' Granda-Osp. Maggiore Policlinico, Milan, Italy, 5Fondazione IRCCS Istituto Nazionale dei Tumori, Milan, Italy, 6Università degli Studi di Milano, Milan, Italy, 7University of California, San Francisco, San Francisco, CA, 8University of Milano, Milan, Italy.
Abstract
Purpose
While chronic lung allograft rejection (CLAD) limits survival after lung transplantation, it is unclear if this syndrome reflects a single biological process. We hypothesized that immune and cell-signaling pathways differ at the spatial level in subtypes of CLAD.
Methods
Representative lung blocks from 8 patients who underwent re-transplantation for CLAD, were retrieved. Four had BOS and four had RAS. Spatial proteomics of CD45, CD68, and CD20-pos cells was performed using the GeoMX and protein modules for immune-related and cell viability pathways (n=72 proteins) in fibrotic (F; n=18; CD20) and endoalveolar regions (E/A, n=20; CD45, CD68; (Fig. 1A). Significant targets were analyzed at single-cell level using MACSima in re-LT and stable lung tissues and in a published dataset of persistent and progressive acute lung allograft dysfunction.
Results
RAS lungs show greater apoptosis and less proliferation compared to BOS (Fig. 1B) and the immunological state in two CLAD types are globally different (Fig. 1C). Activated cytotoxic (CD8/OX40Lpos) lymphocytes are enriched in RAS fibrotic areas together with memory cytotoxic cells (CD8/CD45RO/PD1pos; Fig. 1D,E). In BOS there is an increase of suppressive exhausted regulatory T cells (CD4/CD25/FOXP3/Tim-3pos) especially in the E/A space (Fig. 1F), together with activated cytotoxic cells (CD3/CD8/GZMA/4-1BBpos). Single-cell analysis showed cytotoxic co-inhibitory receptors, such as Tim-3, to be decreased in RAS lungs (Fig. 1G). Lastly, BOS E/A regions are enriched in CD20-positive B cells and monocyte-derived macrophages (CD14/CD68/CD11cpos), characteristic of an active inflammatory state.
Conclusion
Our spatially resolved approach reveals different lymphocyte and monocyte cell types in BOS and RAS. BOS lungs show predominant expression of immune checkpoints with a suppressive function both in T-lymphocytes and in monocyte cells within E/A space. These immunological differences suggest optimal therapies may differ across CLAD subtypes.