The 2026 ISHLT Consensus Statement on Clinical Cardiac Xenotransplantation
Published 20 February 2026
Mandeep Mehra, MD, MSc; Muhammad Mohiuddin, MD, DSc; Bruno Reichart, MD, PhD; Jan-Michael Abicht, MD; Jacinthe Boulet, MDCM, MPH; David Cooper, MD, PhD; Peter Cowan, PhD; Maria Crespo-Leiro, MD, PhD; Joachim Denner, PhD; Jay Fishman, MD; Bartley Griffith, MD; Wayne Hawthorne, MD, PhD; Emer Joyce, MD, PhD; Igor Konstantinov, MD, PhD; Anuradha Lala, MD; Matthias Langin, MD; Robert Padera, Jr., MD, PhD; Richard Pierson III, MD; Angelika Schnieke, PhD; Linda Scobie, PhD; Patricia Uber, PharmD; Eckhard Wolf, DVM; Ann Woolley, MD, MPH
J Heart Lung Transplant. February 2026.
The global shortage of donor hearts remains one of the most stubborn barriers in advanced heart failure care. Two decades after ISHLT first addressed xenotransplantation, this consensus statement provide a disciplined, evidence-based roadmap for what comes next.
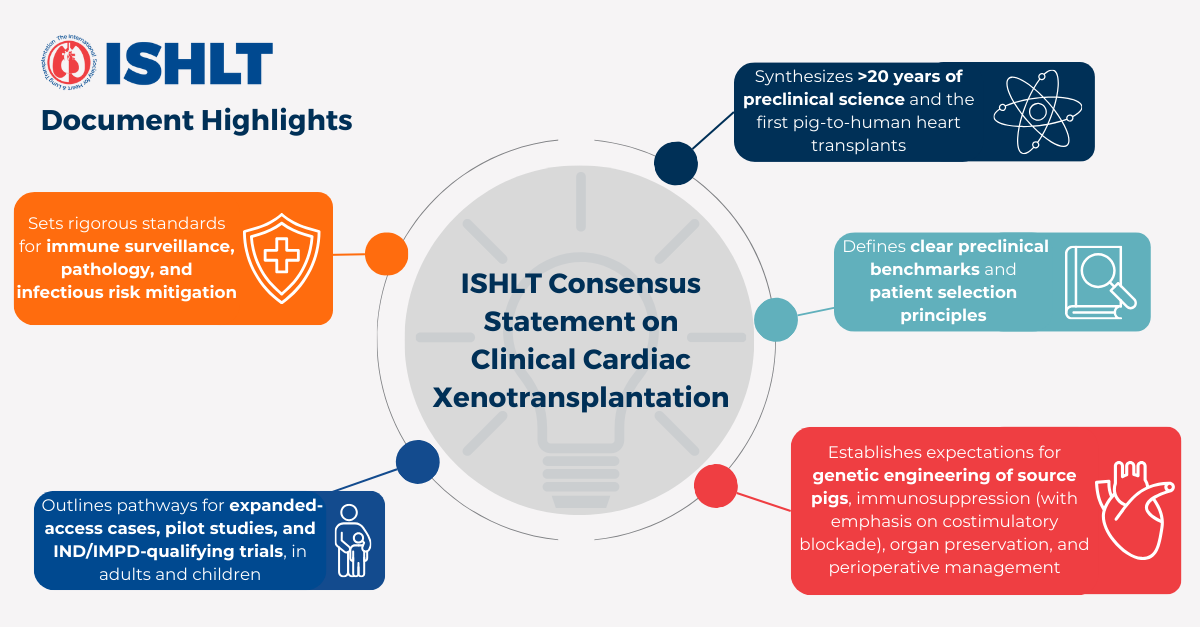
Developed by a multidisciplinary panel of global experts as an update to 2000 ISHLT statement on xenotransplantation, this document reflects more than two decades of scientific progress, including the first two pig-to-human heart transplant experiences, and provides a structured framework addressing:
- Minimum preclinical evidence required before human trials
- Criteria for selecting appropriate adult and pediatric candidates
- Required genetic modifications of donor pigs
- Standards for surgical technique, perioperative care, and immunosuppression
- Expectations for immune monitoring, pathology, and infection surveillance
- Regulatory oversight and international coordination
This consensus statement is intended as a living blueprint, to guide clinicians, investigators, regulators, and industry partners as cardiac xenotransplantation enters its next, more disciplined phase.
Read at JHLTRead the Perspective Piece at JHLT Download the Perspective Piece
The ISHLT Perspective Piece is a new document type developed to accompany all ISHLT Documents and is specifically designed for print publication in JHLT. The piece The piece will set the context for the document in an easy-to-read manner and serves as an opportunity for writing groups to speak directly to the heart and lung transplant community about the rationale behind each document, its most important takeaways, and the hard work that went into creating it.
Top Takeaways
Click to view larger image and all document highlights.
Related Guidlines
-
HFSA/SAEM/ISHLT Clinical Expert Consensus Document on the Emergency Management of Patients with Ventricular Assist Devices
-
Present Status of Research on Psychosocial Outcomes in Cardiothoracic Transplantation— Review and Recommendations for the Field
-
Report from a Consensus Conference on Primary Graft Dysfunction after Cardiac Transplantation
-
2019 Updated Consensus Statement on the Diagnosis and Treatment of Pediatric Pulmonary Hypertension
-
The 2015 ISHLT Guidelines for the Management of Fungal Infections in Mechanical Circulatory Support and Cardiothoracic Organ Transplant Recipients: Executive Summary

